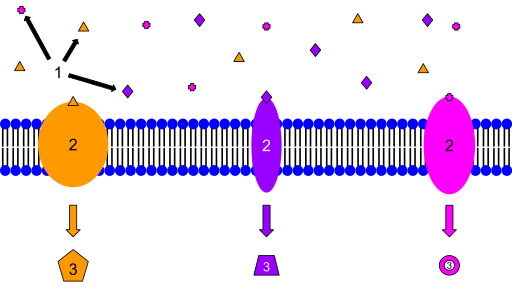
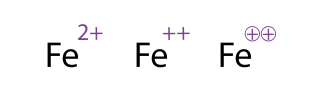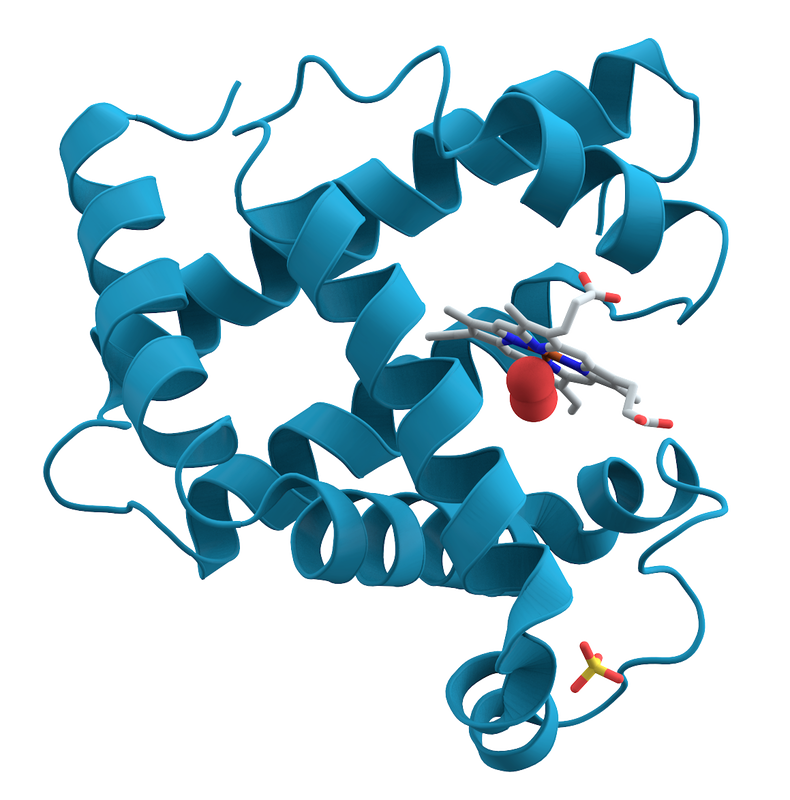Chemistry and The Chemical Level of Organization:
- The human body consists of atoms, molecules and compounds at its most basic level of organization
- Chemistry: the study of matter and its components and interactions
The Biochemistry of the Human Body:
Matter:
Matter is that which takes up space and has mass. It exists in many different forms, including liquids, solids, gases, oils, and metals. Matter is composed of chemical elements in pure form and in combinations (compounds).
Mass is the amount of matter found in an object. The weight of an object is basically a measurement of its mass.
Weight is how strongly mass is pulled by gravity. Weight is basically a measurement of an object's mass.
The human body is about 76% water by weight and each cell consists of about 90% water with dissolved nutrients called cytoplasm or cytosol.
Mass is the amount of matter found in an object. The weight of an object is basically a measurement of its mass.
Weight is how strongly mass is pulled by gravity. Weight is basically a measurement of an object's mass.
The human body is about 76% water by weight and each cell consists of about 90% water with dissolved nutrients called cytoplasm or cytosol.
Element:
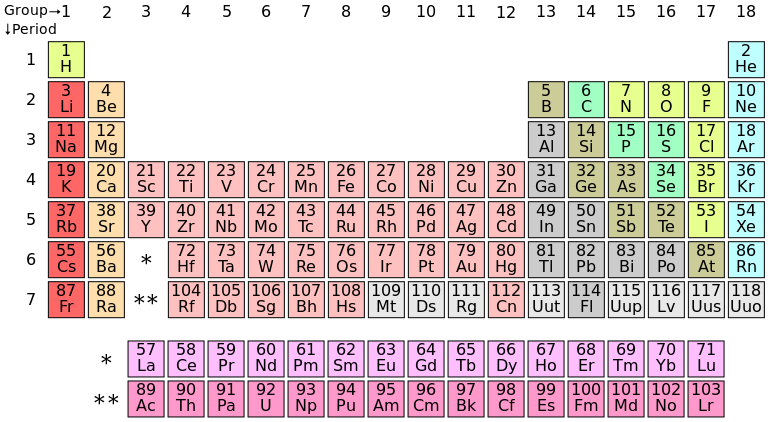
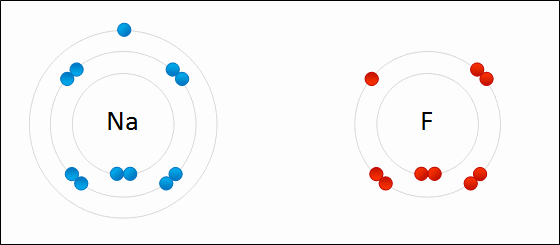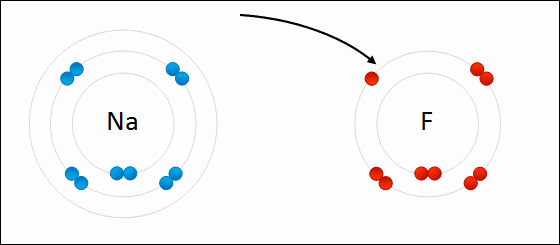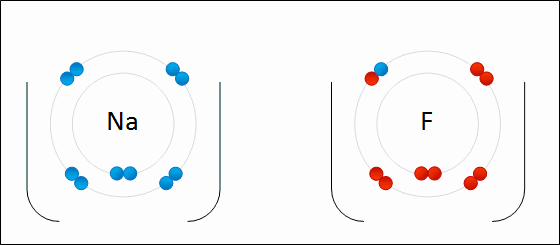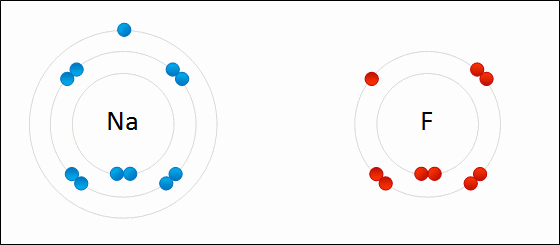
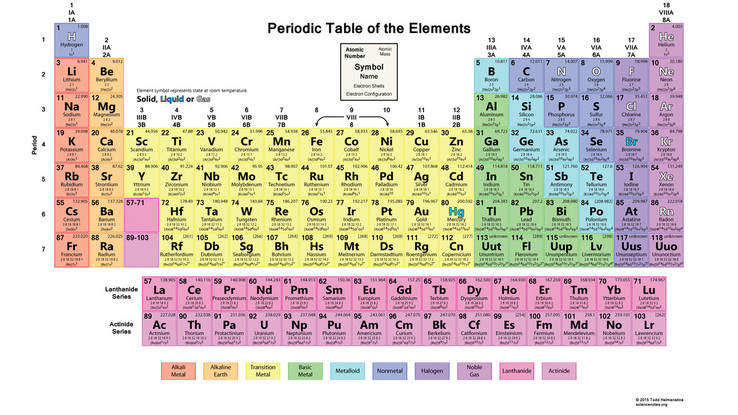
Elements are substances that cannot be further broken down into other substances by chemical or metabolic reactions. Matter is composed of elements. Currently, there are 92 known elements, but scientists are discovering new ones all the time. Each element has a symbol, as seen in the periodic table of elements. Each symbol is typically the first 1-2 letters of the element's name, but some symbols are named from Latin or German terms.
What makes each element unique? Each element consists of specific types of atoms different from the atoms of other elements.
A. ESSENTIAL ELEMENTS:
B. MINERALS
C. TRACE ELEMENTS:
What makes each element unique? Each element consists of specific types of atoms different from the atoms of other elements.
A. ESSENTIAL ELEMENTS:
- Comprise about 1/4th-1/5th of the 92 natural elements
- Needed for life and reproduction
- Humans need 25 of these elements to sustain life
- Plants need 17 of these
- Oxygen (O), Carbon (C), Hydrogen (H) and Nitrogen (N) comprise approximately 96% of living matter
- Calcium (Ca), Phosphorus (P), Potassium (K), Sulfur (S) and others comprise about 4% of the mass of an organism
B. MINERALS
C. TRACE ELEMENTS:
- Required only in tiny, minute amounts
- Iron (Fe)
- Iodine (I)
- Sodium (Na)
- Chlorine (Cl)
- Magnesium (Mg)
- Boron (B)
- Chromium (Cr)
- Cobalt (Co)
- Copper (Cu)
- Fluorine (F)
- Manganese (Mn)
- Molybdenum (Mo)
- Selenium (Se)
- Silocon (Si)
- Tin (Sn)
- Vanadium (V)
- Zinc (Zn)
The Periodic Table of Elements:
Compound:
A compound is a substance that is composed of 2 or more different elements. Examples:
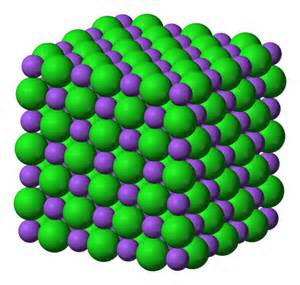
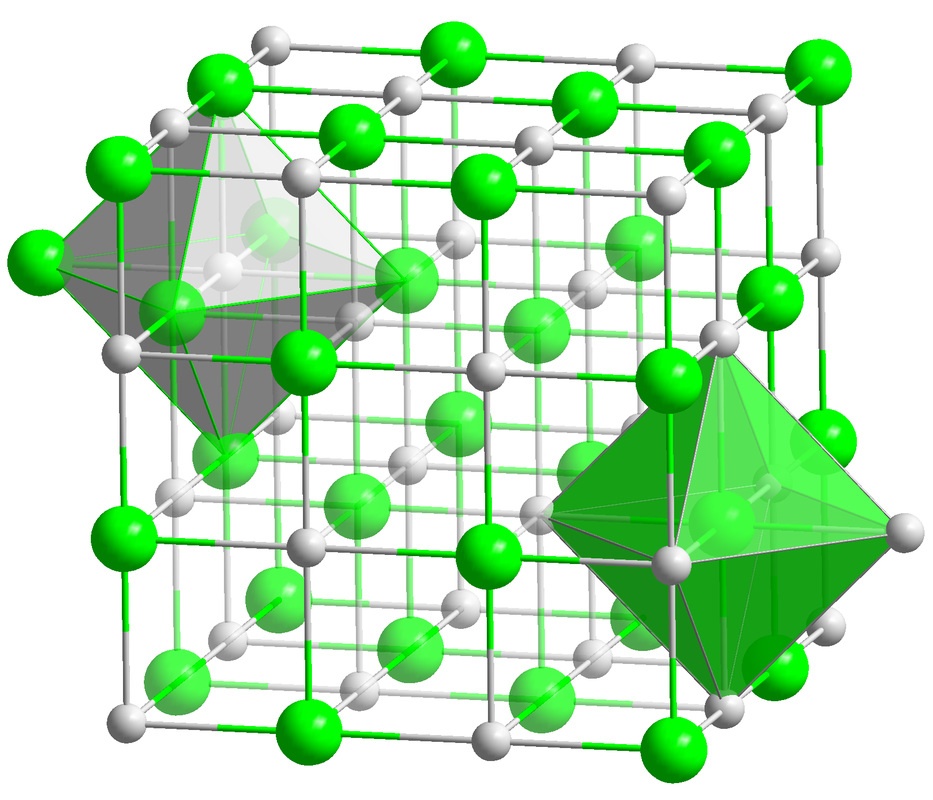
- NaCl (Sodium Chloride)-Table Salt, is a compound composed of a 1:1 ratio of sodium (Na) and chlorine (Cl), a combination of a metal and a gas chemically combined and bonded together to form an edible compound.
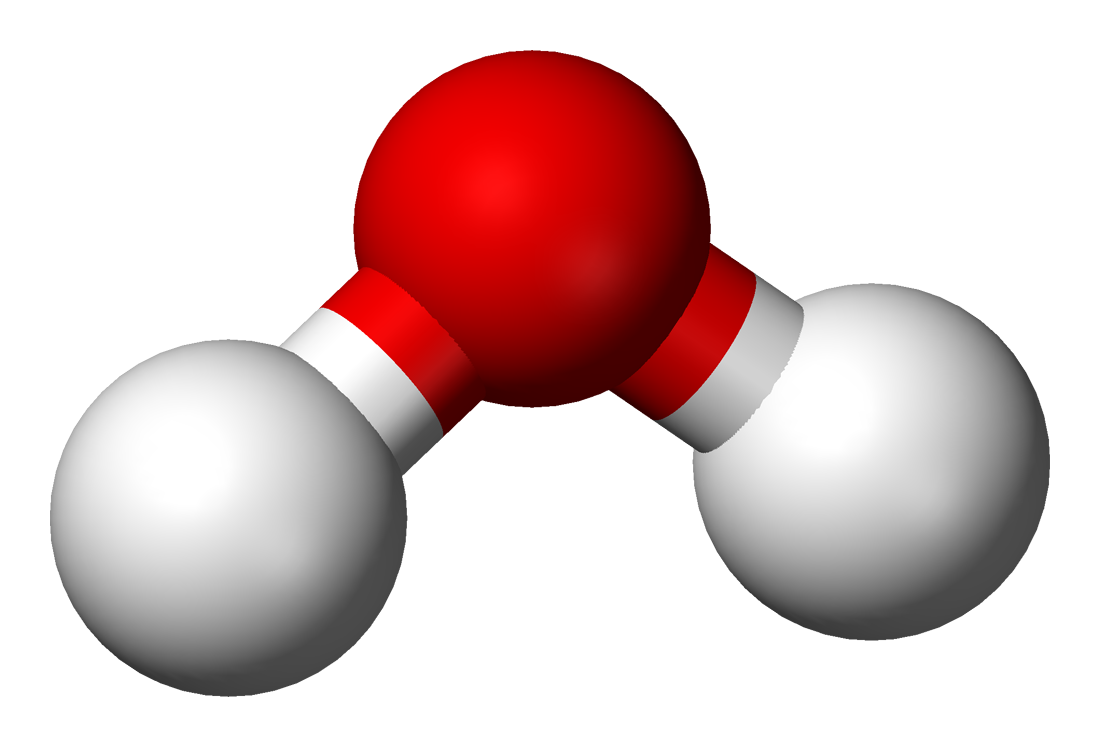
- H2O (Water)-Water is a compound consisting of the 2:1 ratio of the elements hydrogen (H) and oxygen (O).
SUBATOMIC PARTICLES:
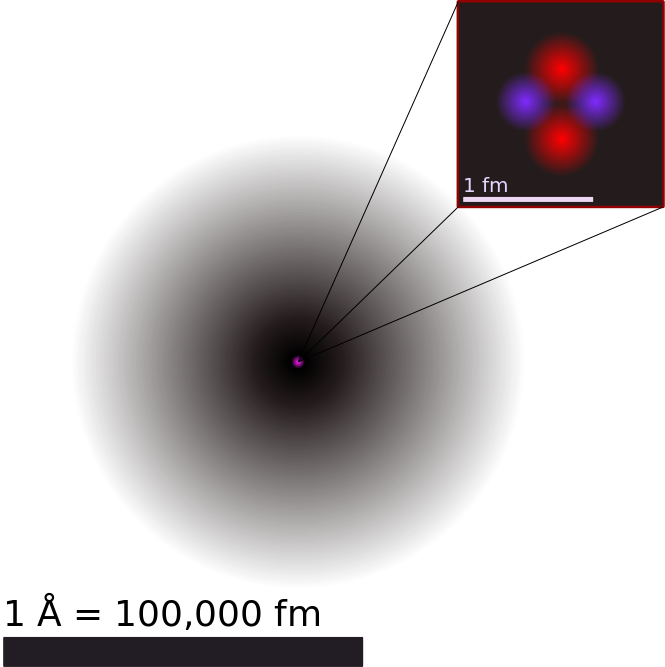
The Atom:
The atom is basically the tiniest unit of matter that keeps the properties of an element, making each element unique. Atoms are composed of tiny subatomic particles: protons, neutrons and electrons.
Protons, Neutrons and Electrons:
Electron Shell Model:
Space-Filling (Electron Cloud) Model:
Protons and electrons have an electric charge, whereas neutrons are neutral and do not.
A. PROTONS
A. PROTONS
- Has one unit of positive (+) charge
- Has one unit of negative (-) charge
- Level of energy correlates with its average distance from the nucleus of an atom
- Found in different electron shells, which each possess an average energy level and distance
- The first electron shell is closest to the nucleus and the electrons here possess the lowest potential energy
- Electrons in the second shell possess more potential energy
- Electrons in the third shell possess even more potential energy
- Electrons change the shell they occupy by gaining or losing an amount of energy equal to the difference in potential energy between their position in the old shell and in the new shell, which occurs by the loss or gain of electrons in the shell
- An atom's chemical action depends mainly on the number of electrons in the outermost shell. These electrons are referred to as valance electrons residing in the valance shell
- Neutral: have no charge
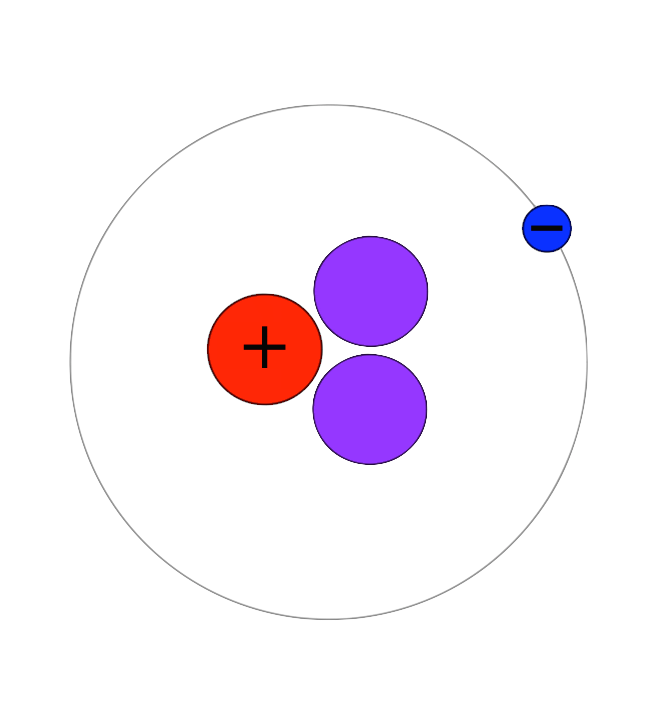
Atomic Nucleus:
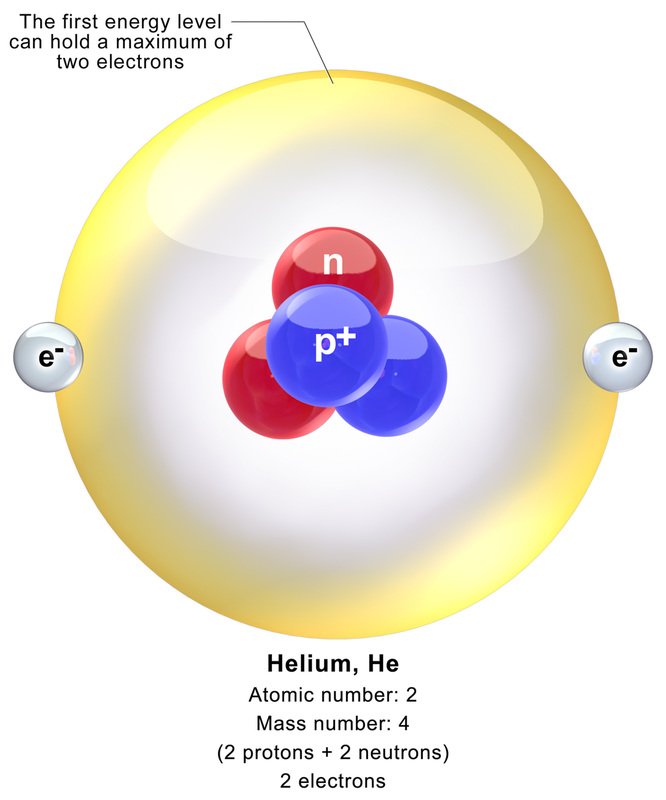
Here, protons and neutrons are packed tightly together in a dense core. This is located at the center of the atom. Protons give the nucleus a positive (+) charge. Electrons orbit around the nucleus, forming a cloud of negative (-) charge around it, attracting opposite charges. This keeps the electrons balanced and surrounding the nucleus, as seen in the image above and in the images of the helium atom shown in the images below.
The neutron and proton are nearly identical in mass, and are represented by a unit of measurement referred to as the dalton.
The atoms of each element vary in their number of subatomic particles, but all atoms of a specific element have the same number of prtons in their nuclei, unique to that particular element.
The neutron and proton are nearly identical in mass, and are represented by a unit of measurement referred to as the dalton.
The atoms of each element vary in their number of subatomic particles, but all atoms of a specific element have the same number of prtons in their nuclei, unique to that particular element.
Atomic Number and Atomic Mass:
A. ATOMIC NUMBER
The simplest atom is hydrogen. It has NO neutrons. It consists of 1 proton and 1 electron.
- The number of protons unique to a particular element
- Because most atoms are neutral in electrical charge, the protons must be balanced by an equal number of electrons
- Written or notated as a subscript to the left of the symbol for that element
- Allows the reader to know the number of protons AND the number of electrons in an electrically neutral atom
- The number of neutrons + protons in the nucleus of an atom
- Written as a subscript to the left of the element's symbol
- The TOTAL MASS of an atom (in daltons)
- Includes protons + neutrons + electrons
The simplest atom is hydrogen. It has NO neutrons. It consists of 1 proton and 1 electron.
Isotopes:
Isotopes are different atomic forms of the same element. This is due to the fact that all atoms of the given element contain the SAME number of PROTONS, but some atoms have more neutrons than others of the same element. This results in different masses.
Carbon (C) is an example of an isotope. There are three isotopes of the element Carbon.
A. STABLE ISOTOPES
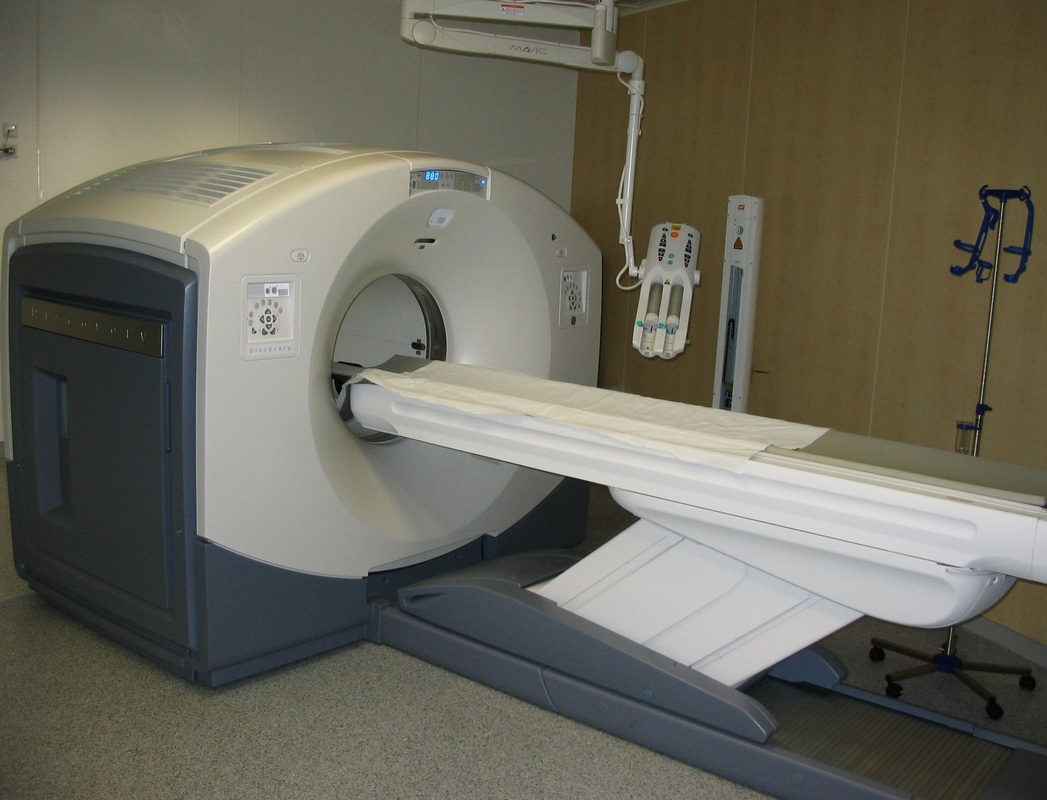
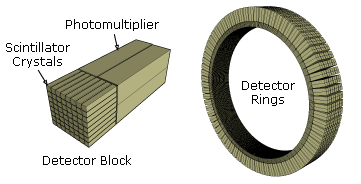
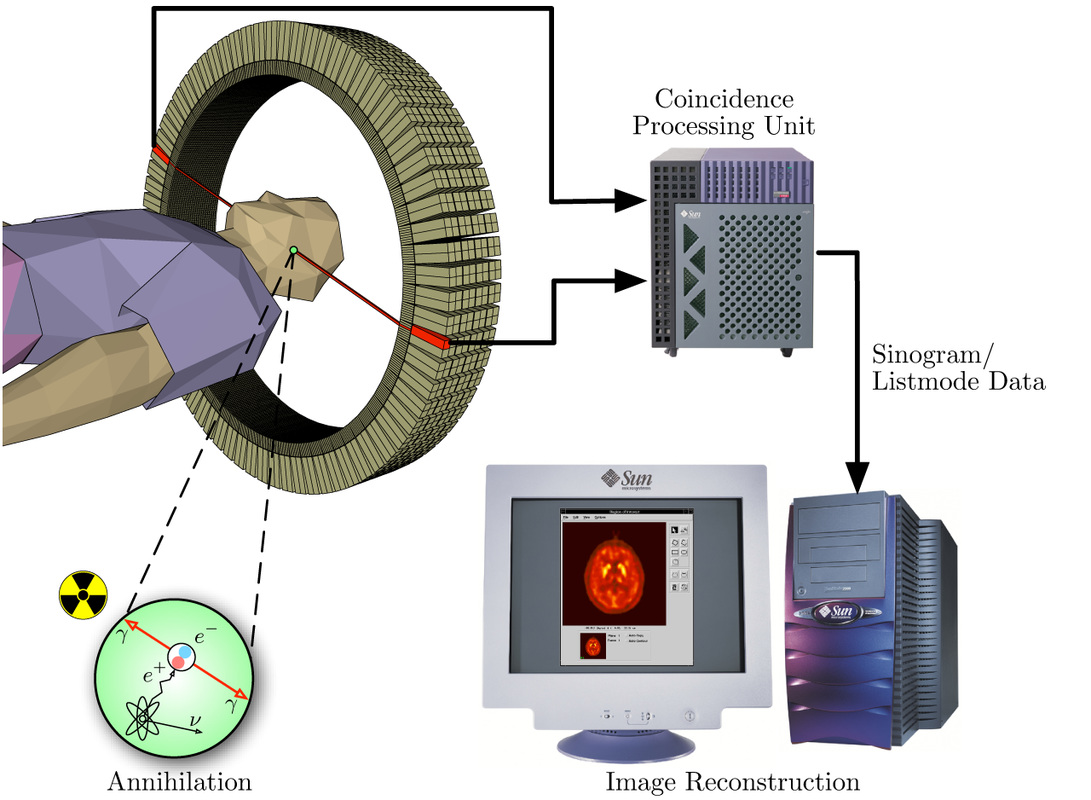
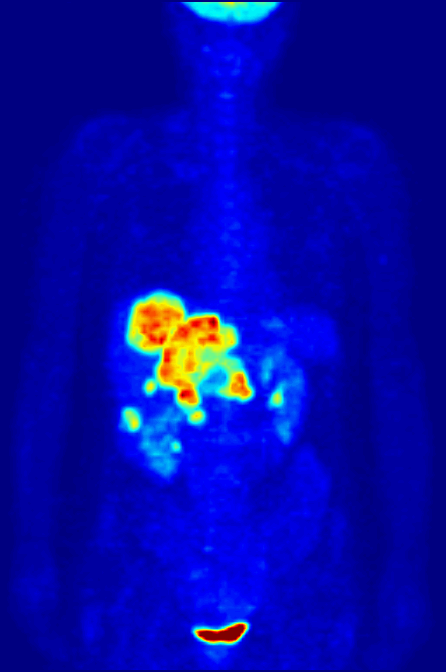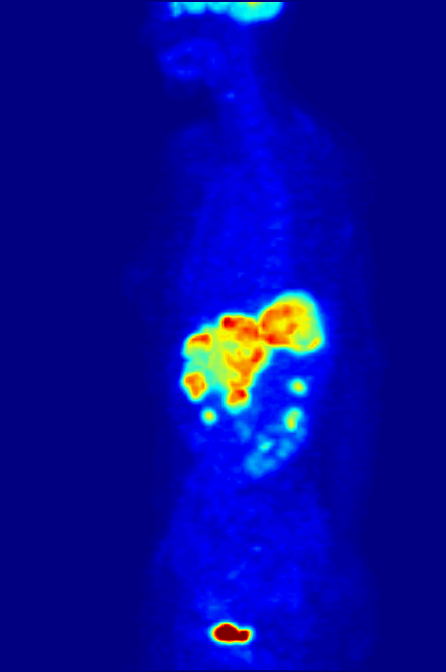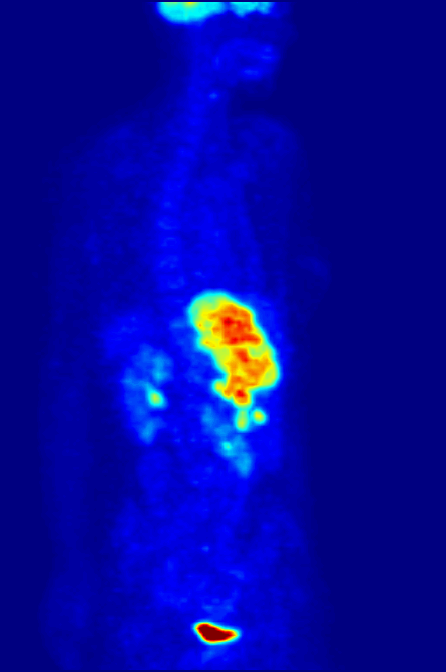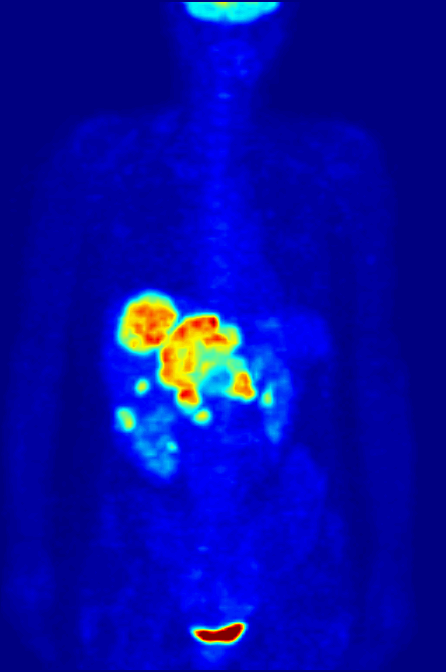
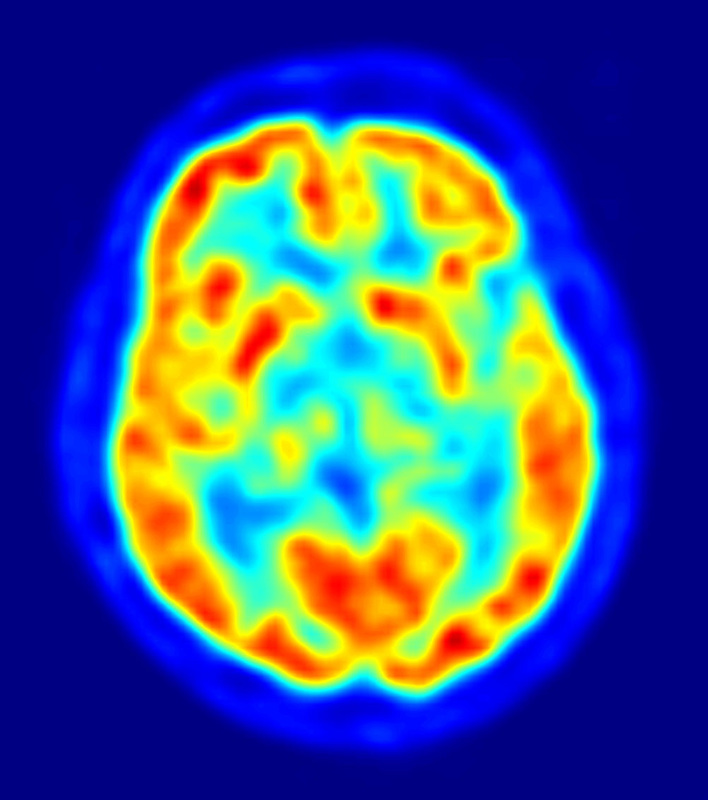
PET scans stand for "Positron-Emission Tomography". This scan is used in medicine to detect locations of intense or strong chemical activity in the body. The bright areas indicate areas of elevated radioactivity, which are labeled with radioactive isotopes, indicating high metabolic activity, which is a hallmark sign of cancer or abnormalities in tissue. This scan is useful for detecting various types of cancers, kidney stones, thyroid goiters, physiologic anomalies, and changes in vasculature (ex: strokes, blockages, plaques due to Alzheimer's Disease).
Carbon (C) is an example of an isotope. There are three isotopes of the element Carbon.
A. STABLE ISOTOPES
- Their nuclei do not tend to lose particles so they are stable
- Their nucleus may decay spontaneously, releasing particles and energy
- Unstable nucleus
- Radioactive
- Decay leads to a change in the number of protons
- Results in transformation of the atom to an atom of a different element
- Example: Radioactive Carbon (C) decays and becomes Nitrogen (N)
- Used to date fossils (Carbon dating)
- Used as tracers to follow atoms through metabolism (Radioactive tracers), which are readily detected and important diagnostic tools in the field of medicine (kidney pyelograms, PET scanners to detect cancerous growth) (Radioactive iodine, Radioactive fluorine, Radioactive glucose)
- May be hazardous (radioactive fallout from nuclear accidents)
PET scans stand for "Positron-Emission Tomography". This scan is used in medicine to detect locations of intense or strong chemical activity in the body. The bright areas indicate areas of elevated radioactivity, which are labeled with radioactive isotopes, indicating high metabolic activity, which is a hallmark sign of cancer or abnormalities in tissue. This scan is useful for detecting various types of cancers, kidney stones, thyroid goiters, physiologic anomalies, and changes in vasculature (ex: strokes, blockages, plaques due to Alzheimer's Disease).
Contrast dyes, when injected, or those that are swallowed, such as barium sulfate, block the radiation to various body structures, which change the way the imaging machines work and change the way in which the machine observes your soft tissues, organs or bones. For example, radioactive iodine (isotopes of iodine) and barium-sulfate (elements) cause the radiation to be unable to pass through the tissue and block it, so it appears light or white against the dark background. These types are used for CT-scans and other radiographs, which cause certain areas of the body to appear gray or white against the darker background.
MRI contrast dyes work a little differently. They also differentiate between different tissues, however, it the dyes have a magnetic property about them that are absorbed by some tissues in the body. The most common type of dye is the radioactive isotope of the metal gadolinium. The images are picked up by the scanner, which uses a vacuum of electrons, creating an electromagnetic field. It highlights tiny blood vessels, specific soft tissues, including tears, and organs. They will literally glow against the dark background. They have a heavy molecular weight than other particles due to the metal property. They stay inside the organ system being tested and are unable to diffuse into nearby tissues. If you have ever heard of gamma knife surgery, this type of contrast is used for the surgery, particularly on the brain. It can also be used to look for infections, cancer, metastatic cancer, multiple sclerosis, the pituitary gland for tumors, or neurofibromatosis, bone masses or lesions or infections, abscesses, ulcers, cellulitis, cysts, fibroids, kidney and liver function. Due to filtration, it takes the kidneys about 24 hours to filter it from the body.
Some individuals are allergic to contrast dyes or will experience delayed hypersensitivity allergic reactions, so there are also some signs and symptoms to watch for.
MRI contrast dyes work a little differently. They also differentiate between different tissues, however, it the dyes have a magnetic property about them that are absorbed by some tissues in the body. The most common type of dye is the radioactive isotope of the metal gadolinium. The images are picked up by the scanner, which uses a vacuum of electrons, creating an electromagnetic field. It highlights tiny blood vessels, specific soft tissues, including tears, and organs. They will literally glow against the dark background. They have a heavy molecular weight than other particles due to the metal property. They stay inside the organ system being tested and are unable to diffuse into nearby tissues. If you have ever heard of gamma knife surgery, this type of contrast is used for the surgery, particularly on the brain. It can also be used to look for infections, cancer, metastatic cancer, multiple sclerosis, the pituitary gland for tumors, or neurofibromatosis, bone masses or lesions or infections, abscesses, ulcers, cellulitis, cysts, fibroids, kidney and liver function. Due to filtration, it takes the kidneys about 24 hours to filter it from the body.
Some individuals are allergic to contrast dyes or will experience delayed hypersensitivity allergic reactions, so there are also some signs and symptoms to watch for.
Energy:
A. ENERGY
b) CALORIE-the amount of heat needed to raise the temperature of 1 g of water by 1 degree Celsius and the amount of heat that 1 g of water releases when it cools down by 1 degree Celsius
c) KILOCALORIE-1,000 calories; amount of heat needed to raise the temperature of 1 kg of water by 1 degree Celsius
d) Joule (J)-a unit of energy that equals 0.239 calories; 1 calorie = 4.184 J.
D. EVAPORATION
- An atom's electrons vary in their amount of energy
- Defined as work and the capacity to cause transformation by doing work
- Energy that matter possesses
- Electrons of an atom possess this type of energy due to their nuclear arrangement
- Negatively charged electrons are attracted to positively charged nucleus, which requires work to move an electron farther away from the nucleus
- The further away the electron from the nucleus, the more work or energy is required (potential energy)
- The energy of anything that moves; The energy of motion
- Possessed by atoms and molecules since they are always moving and changing
- The faster the movement, the greater its kinetic energy
- HEAT-a measure of matter's total kinetic energy and volume as a result of the motion of its molecules
- TEMPERATURE-a measure of the intensity of heat, which represents the average kinetic energy of molecules, and is nondependent upon volume
b) CALORIE-the amount of heat needed to raise the temperature of 1 g of water by 1 degree Celsius and the amount of heat that 1 g of water releases when it cools down by 1 degree Celsius
c) KILOCALORIE-1,000 calories; amount of heat needed to raise the temperature of 1 kg of water by 1 degree Celsius
d) Joule (J)-a unit of energy that equals 0.239 calories; 1 calorie = 4.184 J.
D. EVAPORATION
- EVAPORATIVE COOLING-the cooling down of the surface of a liquid that remains as liquid evaporates
- EVAPORATION/VAPORIZATION-transformation of a liquid to a gas
Bonds:
A. CHEMICAL BONDS
- How atoms combine to form molecules and ions
- Atoms with incomplete valance shells interact with other similar atoms in a way that compliments or completes its valance shell, either by sharing or transferring valance electrons
- Interactions result in atoms sticking together or held together by chemical attractions called chemical bonds
- The strongest chemical bonds are covalent bonds and ionic bonds
- The weakest chemical bonds are hydrogen bonds and van der Waal forces
- The sharing of a pair of valence electrons by two atoms
- Single Bond-a pair of shared electrons by one single bond
- Double Bond-2 or more electrons needed to complete a valance shell and form a molecule by sharing two pairs of valance electrons via two bonds
- 1) Nonpolar Covalent Bond-Electronegativity results in a strong pull of shared electrons towards the atom, resulting in a bond between 2 atoms of the same element, with equally shared electrons (Examples: H2, O2)
- 2) Polar Covalent Bond-When one atom is bonded to a more electronegative atom, the electrons are not shared equally, resulting in a variance of polarity (Example: H2O); Oxygen is the most electronegative of all the elements
- Two atoms are so unequal in their attraction for valance electrons that the stronger electronegative atom takes an electron away from its partner, resulting in loss and gain of electrons (Example: NaCl)
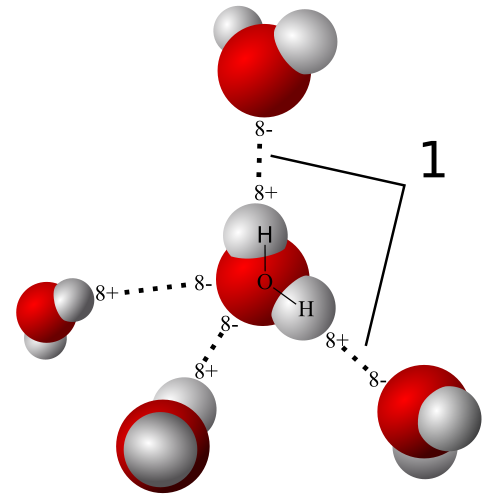
- Partial positive charge on a hydrogen atom
- Noncovalent attraction between a hydrogen atom and an electronegative atom (usually oxygen or nitrogen atoms)
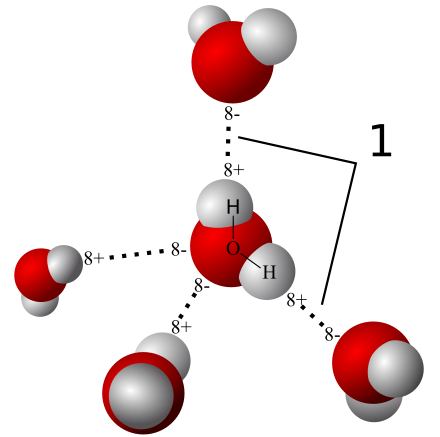
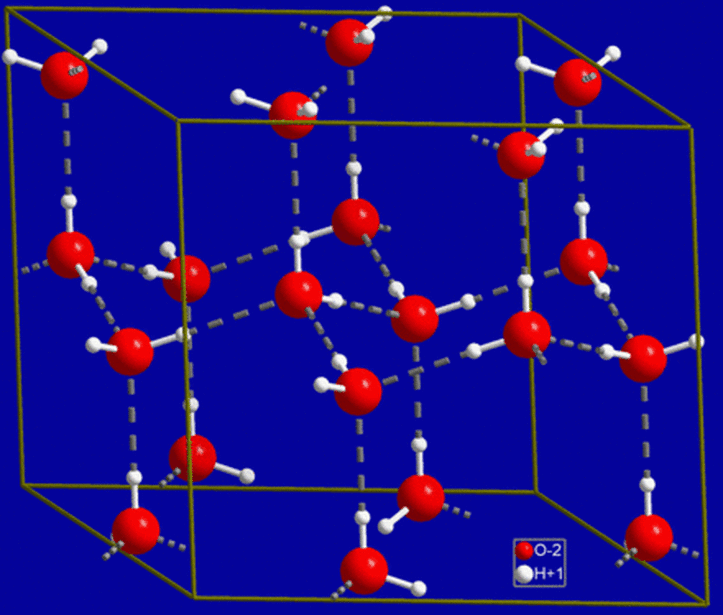
- Most common example is WATER
- Results in COHESION-a collective holding together of a substance by many hydrogen bonds
- Results in ADHESION-the clinging together of one substance to another by hydrogen bonds, which aid in countering the downward pull of gravity
- Results in SURFACE TENSION-a measure of hard it is to stretch or break the surface of a liquid such as water, which possesses a greater surface tension than most other liquids
- Regions or areas of a molecule that are positively and negatively charged, even when they possess nonpolar covalent bonds
- Sticking together of atoms and molecules together as a result of ever-changing regions of positive and negative charge
- Individually weak
- Occur only when atoms or molecules are very close in location to one another
- Many interactions may occur simultaneously, which may be very powerful, resulting in a cumulative effect of reinforcement of a 3D shape of a molecule, such as certain proteins
- The making or breaking of chemical bonds
- Changes in the composition of matter or rearrangement of matter
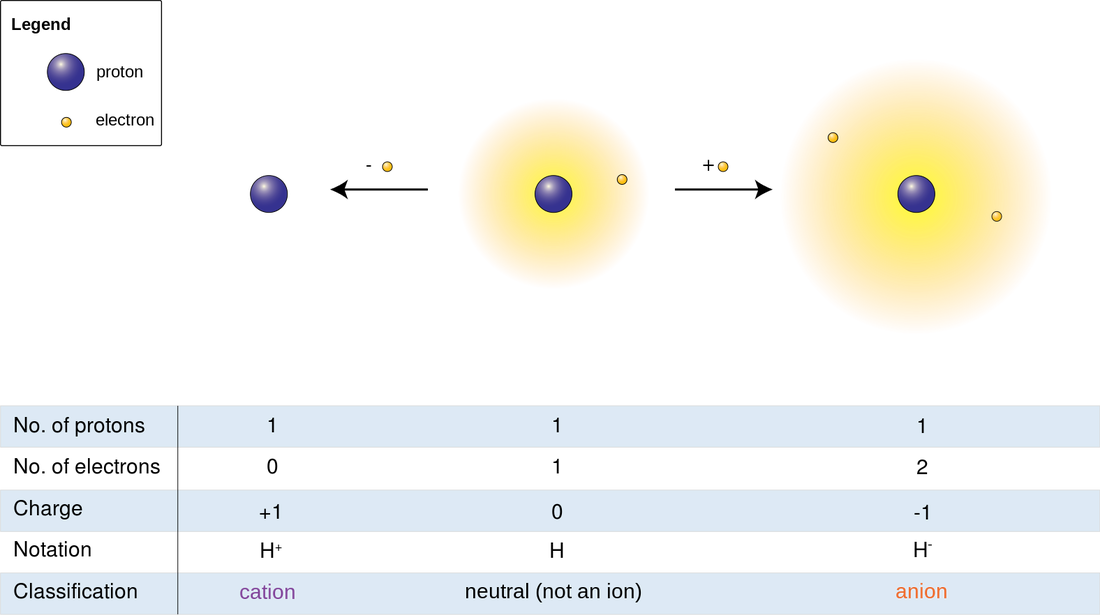
Ions:
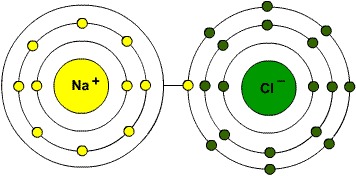
Ions are charged atoms or molecules. Cations are positively charged ions (atom loses electrons), and Anions are negatively charged ions (atom gains electrons). Because of their opposite charges, cations and anions attract each other, resulting in an ionic bond. Transfer of electrons enables a bond to form, resulting in 2 ions of opposite charge.
Compounds formed by ionic bonds are called ionic compounds. These are the salts.
Compounds formed by ionic bonds are called ionic compounds. These are the salts.
Elements:
Molecules:
- Molecules are two or more atoms held together by covalent bonds. Each has a characteristic size and shape and its shape is very important to its function.
- A molecule consisting of 2 atoms is always linear
- Molecules with >2 atoms have more complicated shapes, determined by the position of the atom's orbital (examples: tetrahedron, pyramids with triangular bases, 3D, V-shape)
Compounds:
- Made of atoms of two or more different elements in a fixed ratio, like NaCl (table salt)
Water:
- Solvent-Dissolving agent: There are many substances that are able to dissolve in water, therefore, it is the ideal solvent
- Solute-Substance to be dissolved
- Solution-A liquid that is a complete mixture of 2 ore more substances (Solvent + Solute = solution or mixture)
- Aqueous Solution-Water is the solvent
- Polar ("water-loving")-hydrophilic
- Non-polar ("water-hating")-hydrophobic
- Surface Tension (Water is cohesive, and the molecules tend to stick together)
- Water is adhesive-molecules may form hydrogen bonds with other molecules
- Regulates temperature, including body temperature and climate
- Essential for life
- Liquid, solid, or vapor
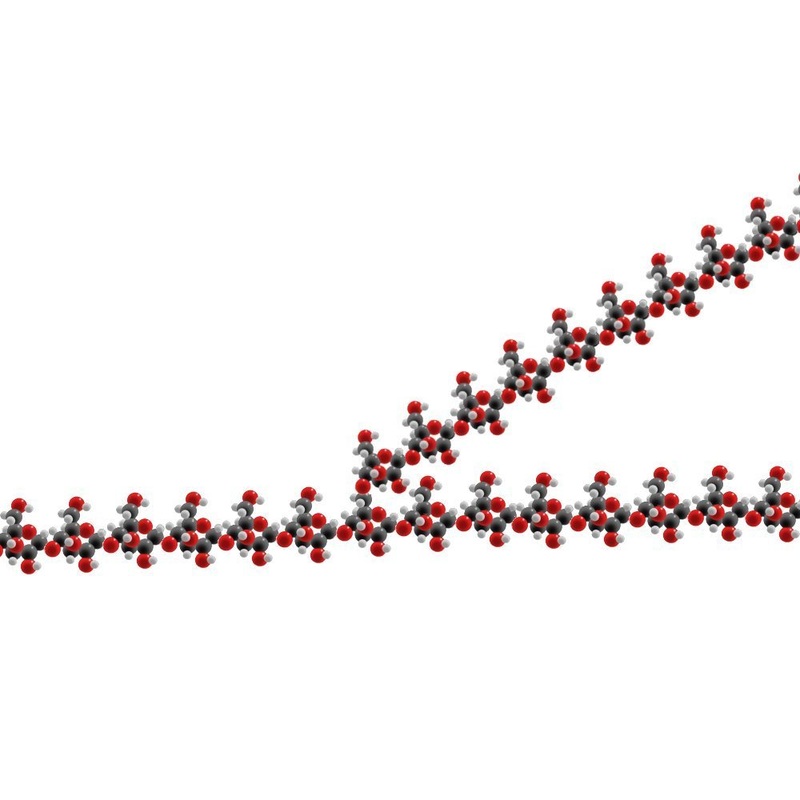
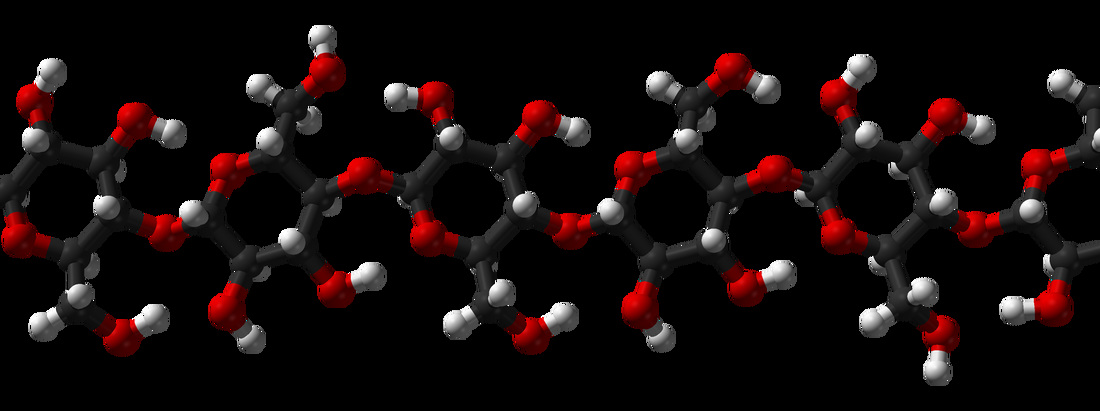
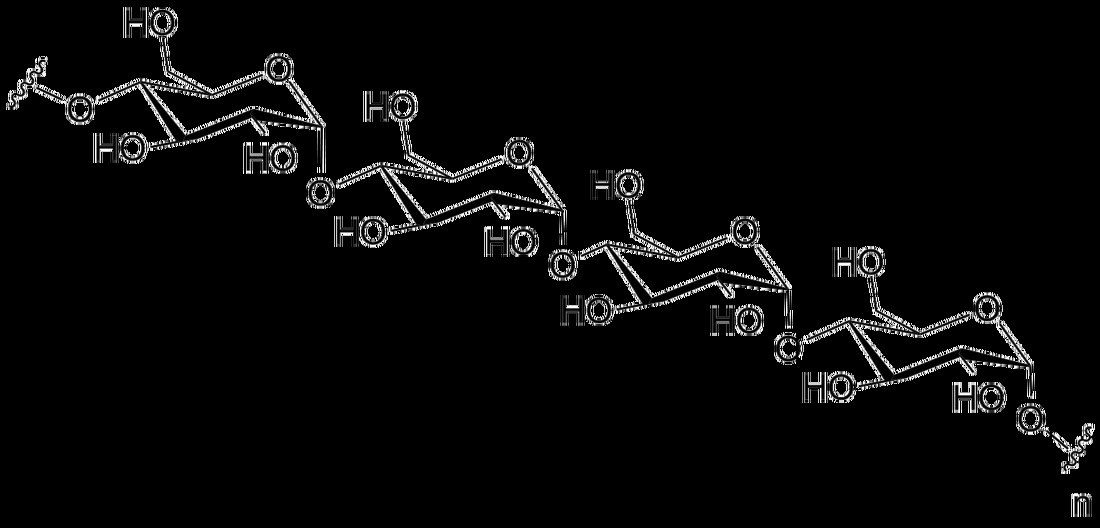
Macromolecules:
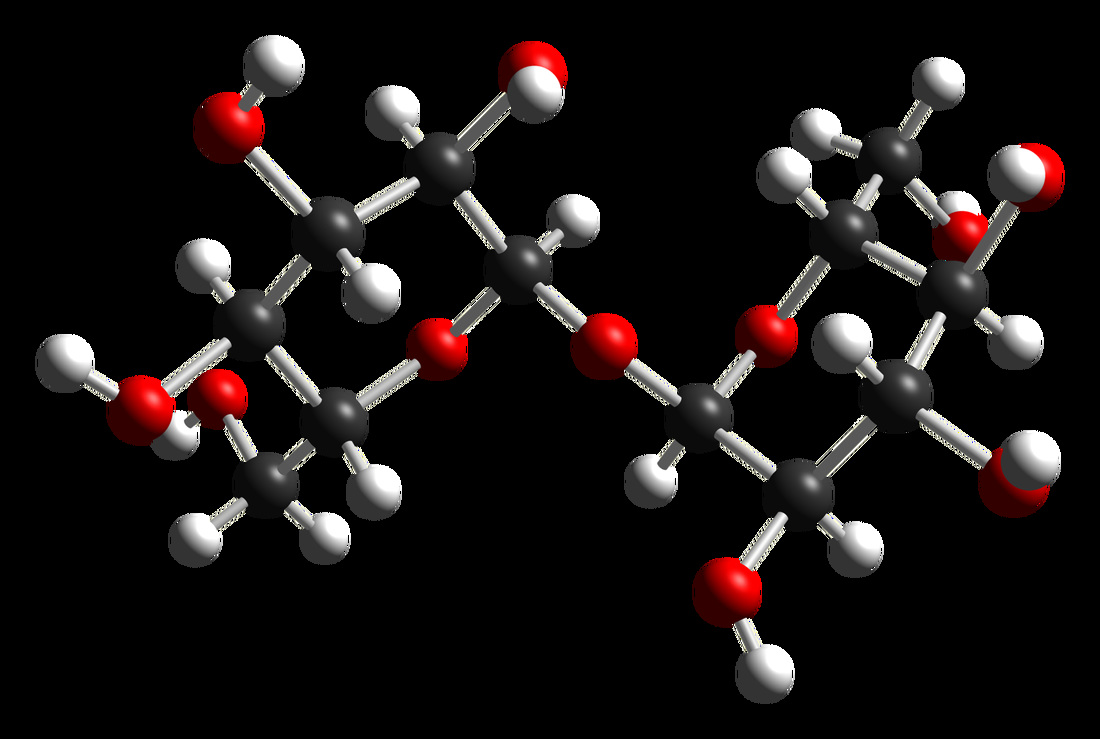
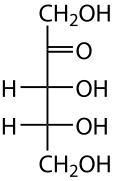
Carbohydrates:
A carbohydrate is a molecule that consists of carbon (C), hydrogen (H) and oxygen (O) atoms.
Saccharide:
A saccharide is another term for a carbohydrate. It comes from the Greek word meaning "sugar", and it includes sugars, starch and cellulose. There are four biochemical groups of saccharides described below. Saccharides typically end in "ose".
1) Monosaccharides:
- Lower molecular weight carbohydrates
- The Simple Sugars
- The simplest carbohydrates that cannot be broken down (hydrolyzed) into smaller carbohydrates
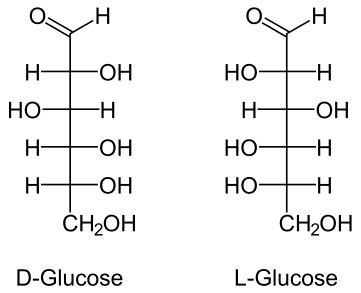
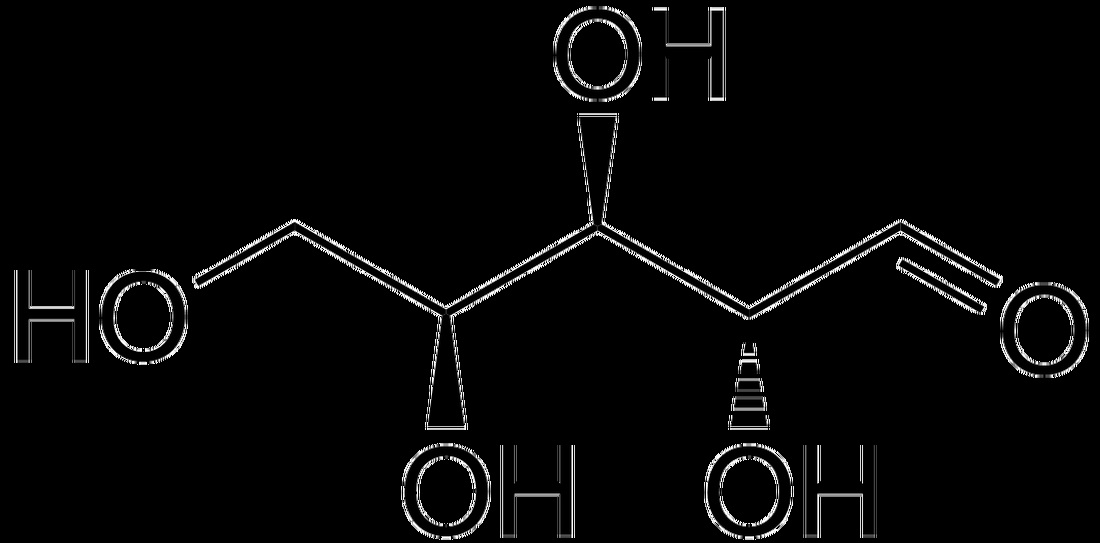
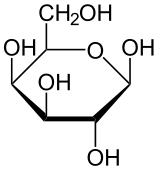
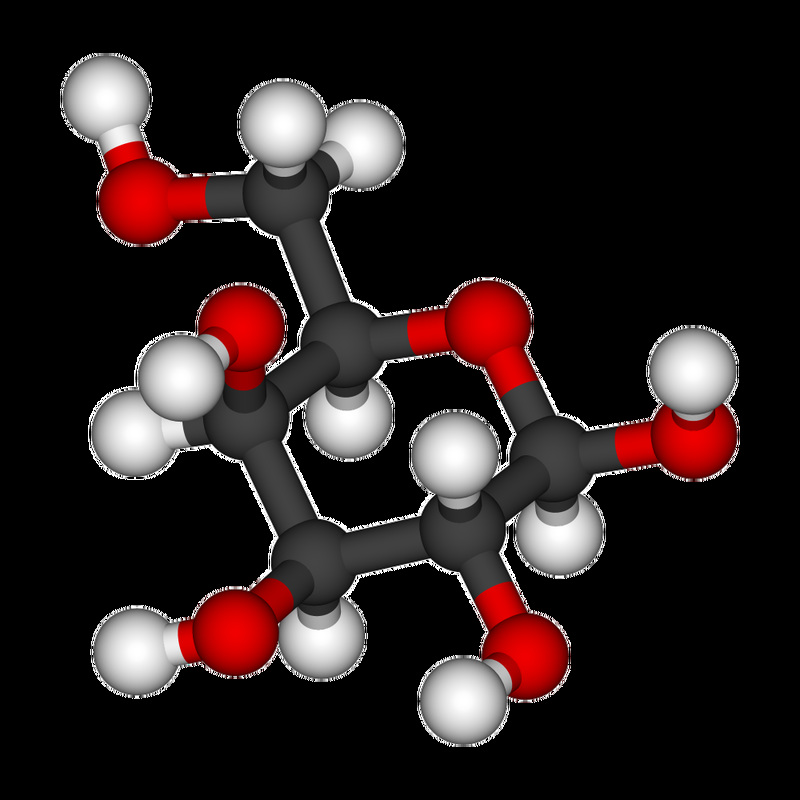
- Glucose
- Aldehydes or ketones with 2 or more hydroxyl groups
- Key fuel molecules for the body and brain and for metabolism (energy and biosynthesis)
- Key molecule in the form of starch for plant metabolic activities
- Building blocks for nucleic acids
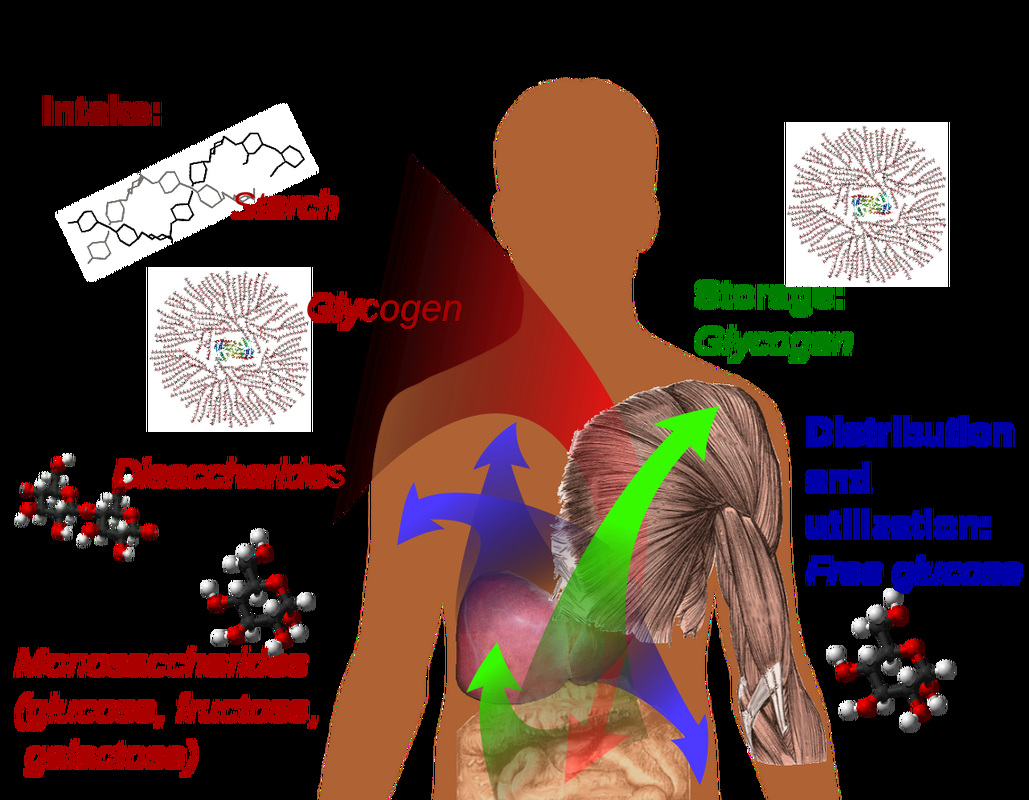
- May be converted to polysaccharides for storage in the body for later use (glycogen), particularly in liver and muscle cells
- Cellulose (most abundant carbohydrate in cell wall of plants and algae)
- Ribose (component of RNA)
- Deoxyribose (component of DNA)
- Lyxose (found in the human heart)
- Galactose (milk sugar)
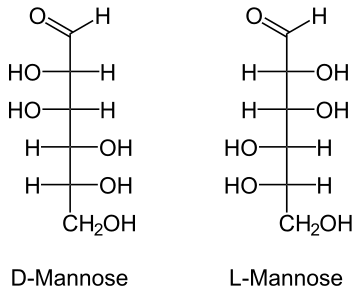
- Mannose (human metabolism)
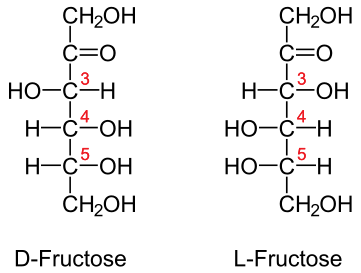
- Fructose (fruit sugar)
- Trehalose (found in insects)
- Ribulose and xylulose (pentose phosphate pathway)
a) Glucose
b) Cellulose
c) Deoxyribose
d) Lyxose
e) Galactose
f) Mannose
g) Fructose
h) Trehalose
i) Ribulose
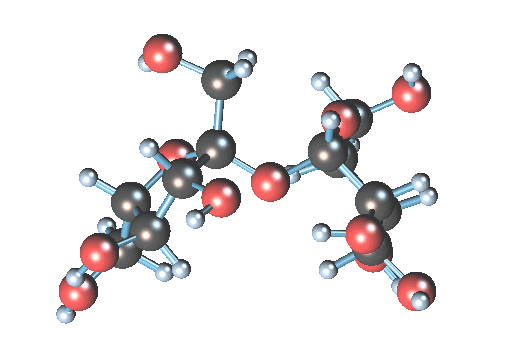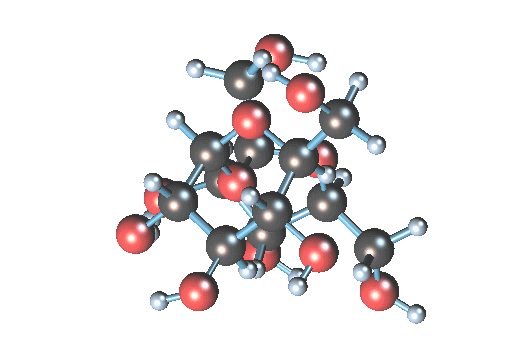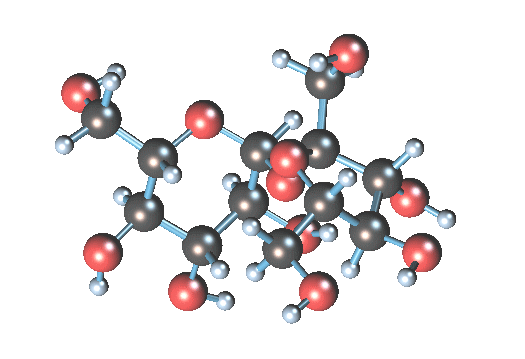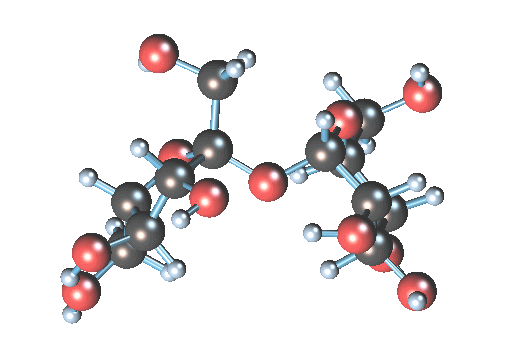
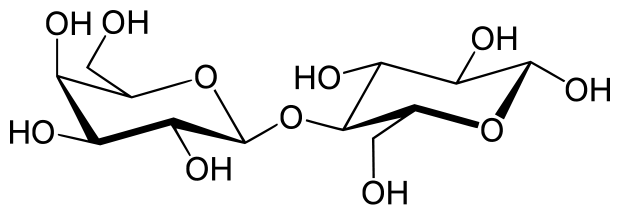
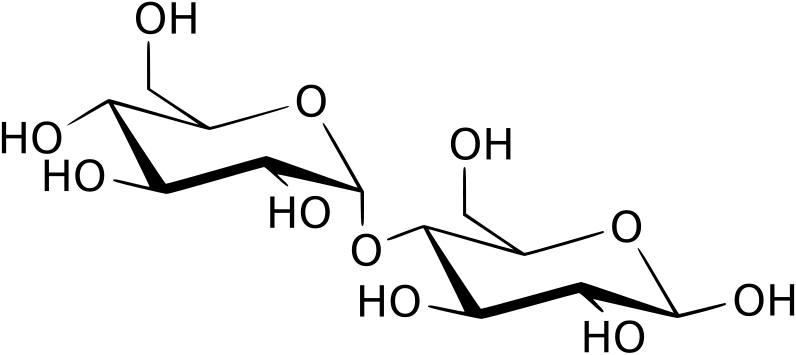
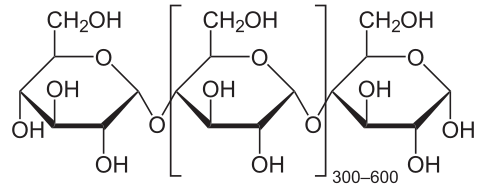
2) Disaccharides:
- Two joined monosaccharides by a glycosydic covalent bond
- Complex
- The simplest polysaccharides
- Sucrose (table sugar)
- Lactose (milk sugar)
- Maltose
a) Sucrose
b) Lactose
c) Maltose
3) Oligosaccharides:
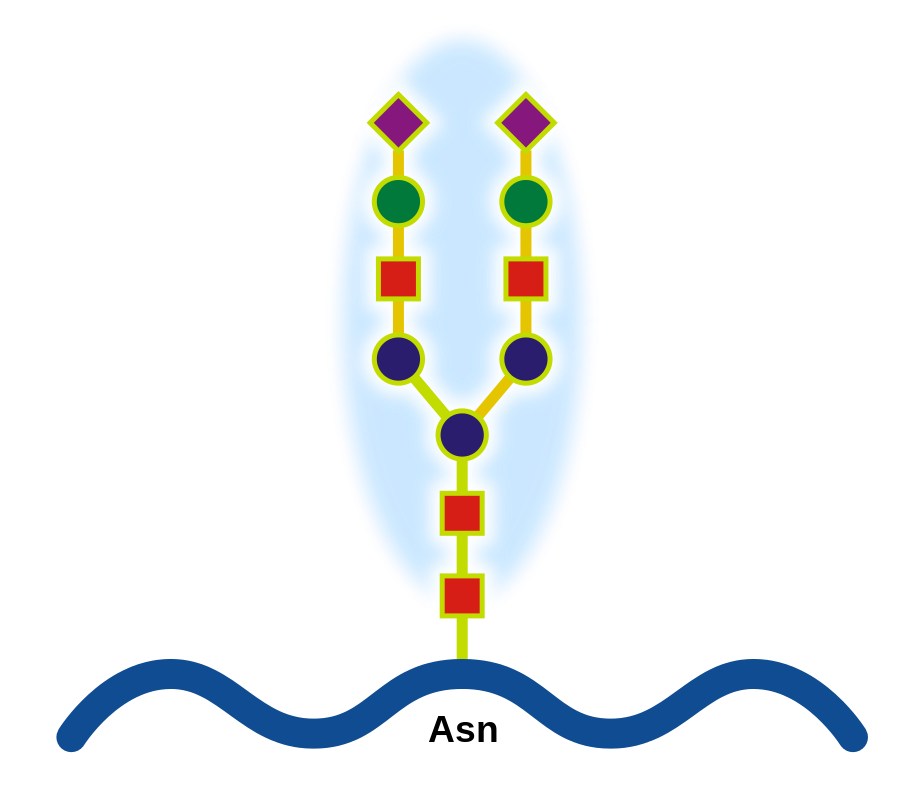
- Contains 3-9 simple sugars linked together
- Found on animal cell membranes as receptors for cell recognition
- Glycoproteins (immunity)
- Glycolipids
- Cell adhesion
a) Glycoproteins
b) Glycolipids
Cell Receptors
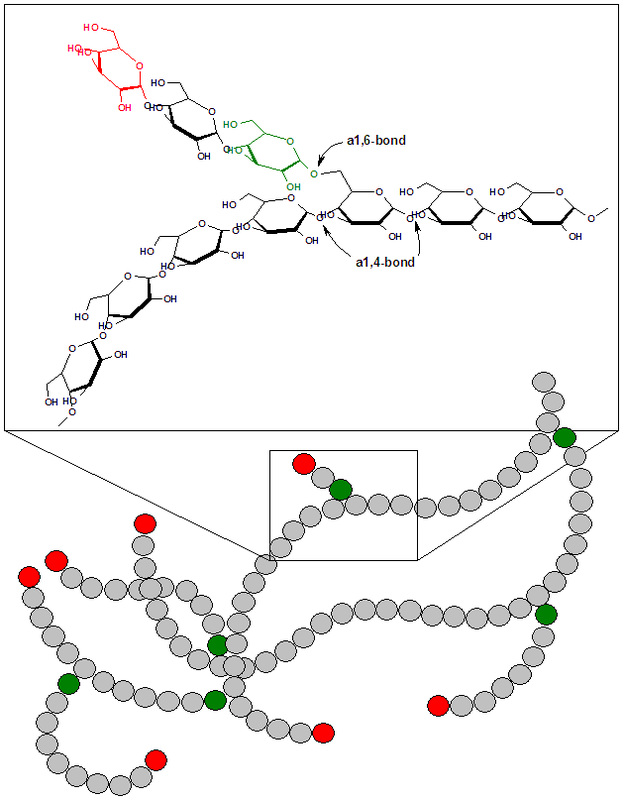
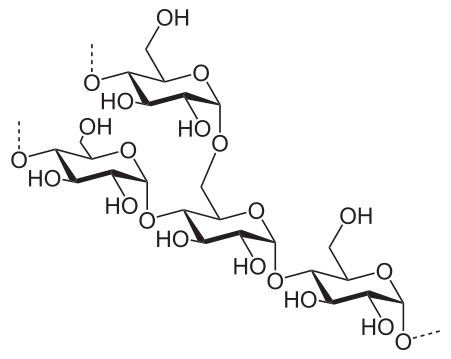
4) Polysaccharides:
- Long chains of monosaccharides
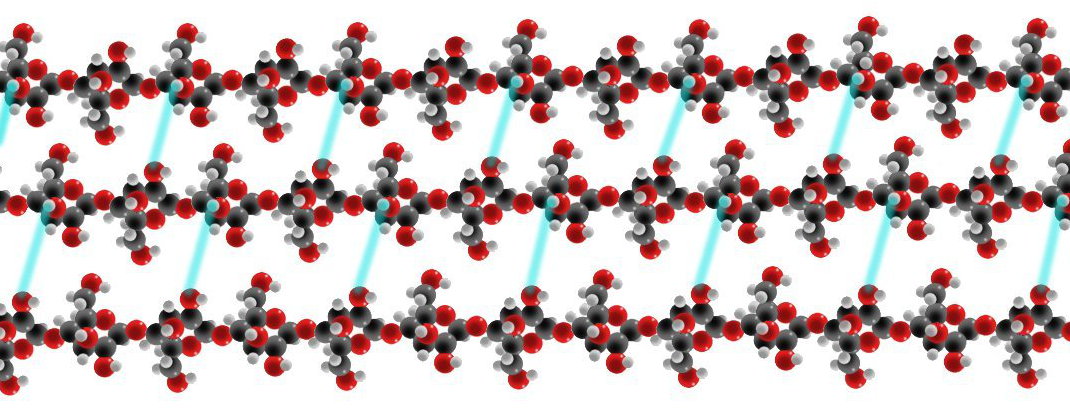
- Starch
- Glycogen
- Cellulose
- Chitin
- Amylose
- Amylopectins/pectins
- Structure
- Storage
- Found in bacterial capsules
- Lipopolysaccharide-found in the cell wall of Gram-negative bacteria
a) Starch
b) Glycogen
c) Cellulose
d) Chitin
e) Amylose
f) Amylopectin
g) Lipopolysaccharide: Bacterial Cell Wall (Gram-negative)
Lipids:
Lipids are molecules that include:
- Fats (A. Unsaturated: 1) Monounsaturated, 2) Polyunsaturated, 3) Trans Fat; B. Saturated; C. Fatty Acid; D. Essential Fatty Acid)
- Sterols (cholesterol)
- Waxes (Beeswax, Lanolin (Carmex, Lip Balms, Lotions/Ointments), Paraffin (Candles, gum, cosmetics, polishes, treatments, Vaseline), Epicuticular-Plants, Carnauba wax-Brazilian tree: shoe polish, wood polish, dental floss, surfboard wax, cosmetics),
- Fat-soluble vitamins (Vitamins D, E, K)
- Phospholipids
- Monoglycerides
- Diglycerides
- Triglycerides
- Store energy
- Provide cellular signaling
- Provide structure for the cell membrane
- Provide membrane fluidity for the cell membrane
- Hydrophobic ("water-hating" or "water-repelling")
- Vesicles for transport
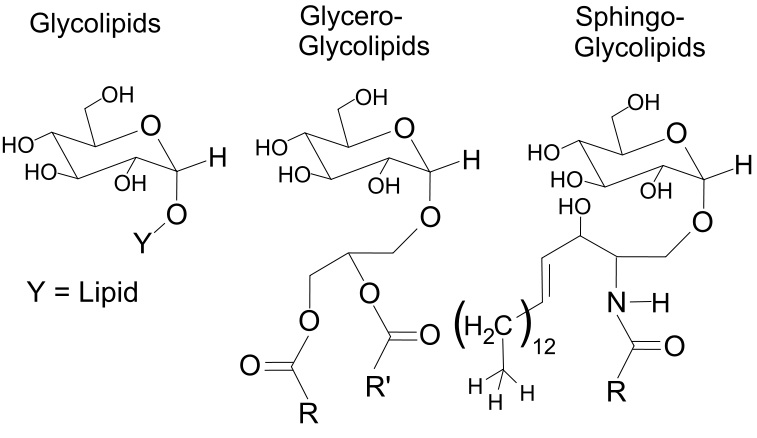
- Glycerolipids
- Glycerophospholipids
- Sphingolipids
- Saccharolipids
- Polyketides
I. Fats
- Provide insulation
- Provide storage
- Provide protection
- Provide energy stores
A. Unsaturated
1). Monounsaturated
2). Polyunsaturated
3). Trans Fat
B. Saturated
C. Fatty Acids
D. Essential Fatty Acids
II. Sterols
- Interconnected carbon rings (4 of them)
A. Cholesterol
B. Testosterone
C. Estrogen
III. Waxes
- Repel water
- Beeswax
- Paraffin wax
- Lanolin
- Fatty acids that are combined with alcohols
IV. Fat-Soluble Vitamins
V. Phospholipids
- Polar/hydrophilic head
- Non-polar/hydrophobic tails
- Provide membrane fluidity for the cell membrane
- Double phospholipid cell membrane
VI. Monoglycerides
VII. Diglycerides
VIII. Triglycerides
- Glycerol head
- 3 fatty acid chains ("tails")
IX. Glycerolipids and Glycerophospholipids
X. Sphingolipids
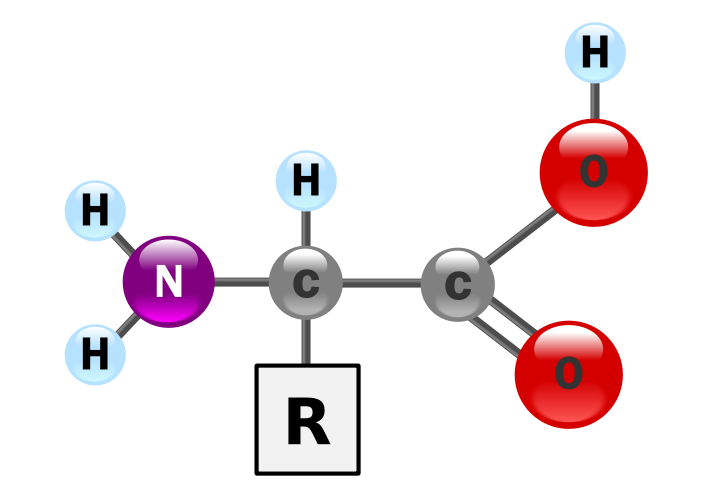
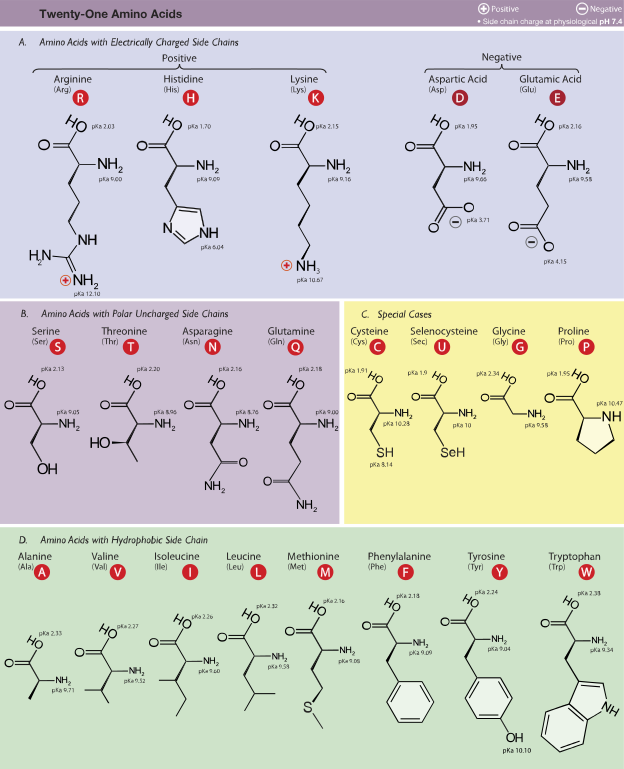
Amino Acids:
Polypeptide:
- Chains of amino acids
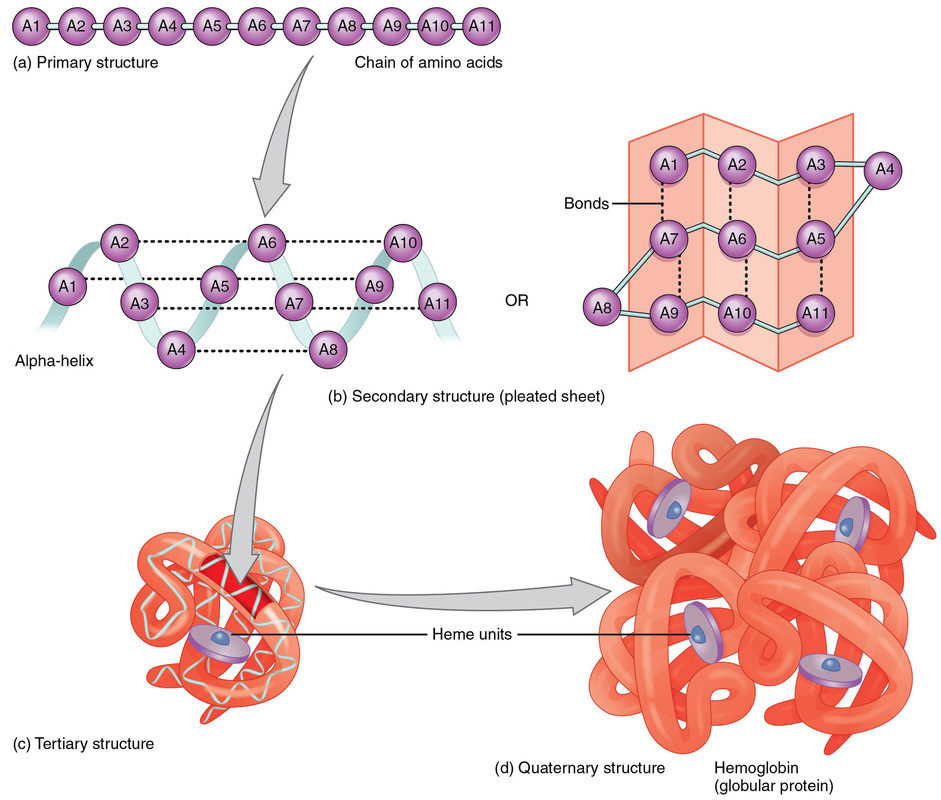
Proteins:
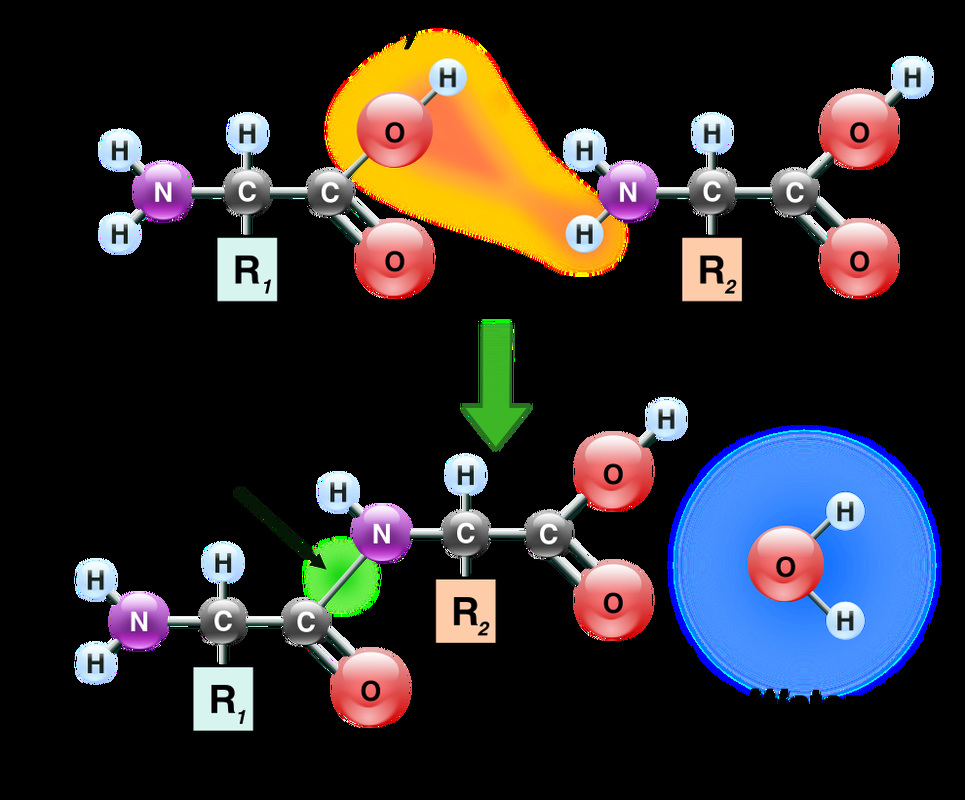
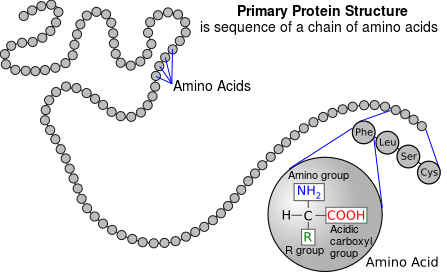
Proteins are made up of 1 or more long chains of amino acid residues bonded together like a pearl necklace.
II. Secondary Structure
III. Tertiary Structure
IV. Quaternary Structure
- Chains of amino acids
II. Secondary Structure
III. Tertiary Structure
IV. Quaternary Structure
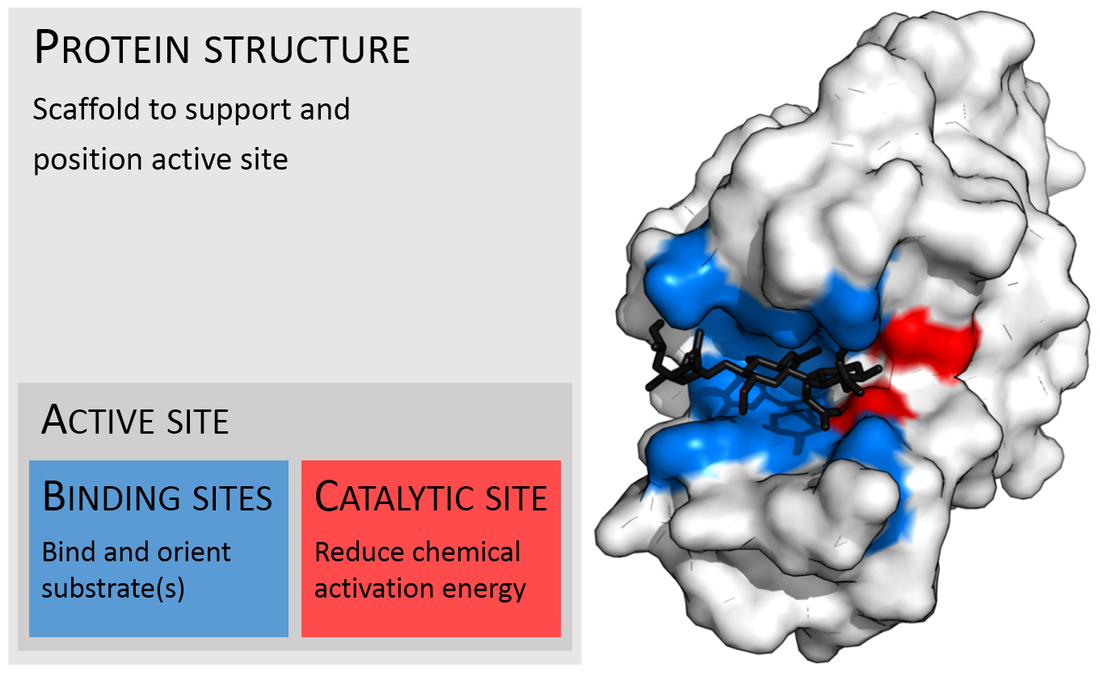
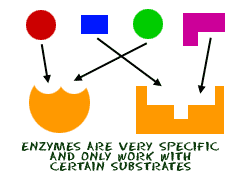
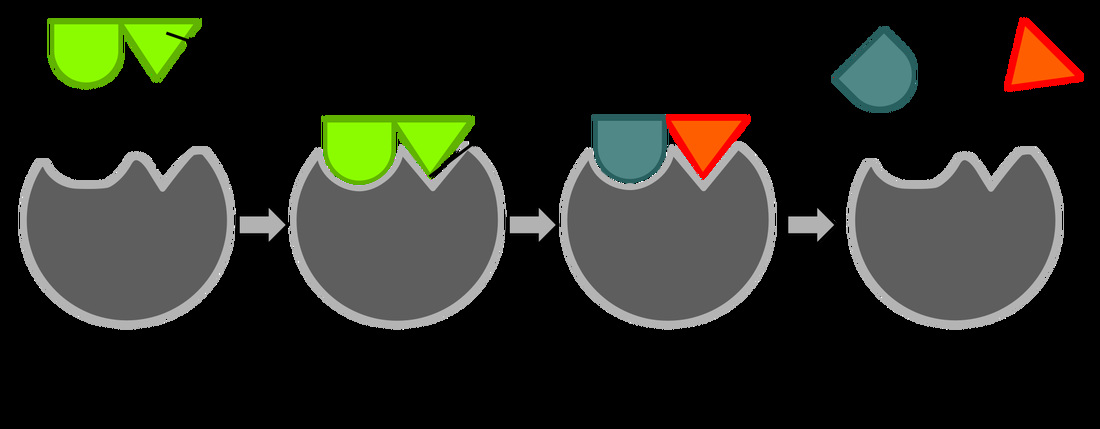
Enzymes:
- Specific
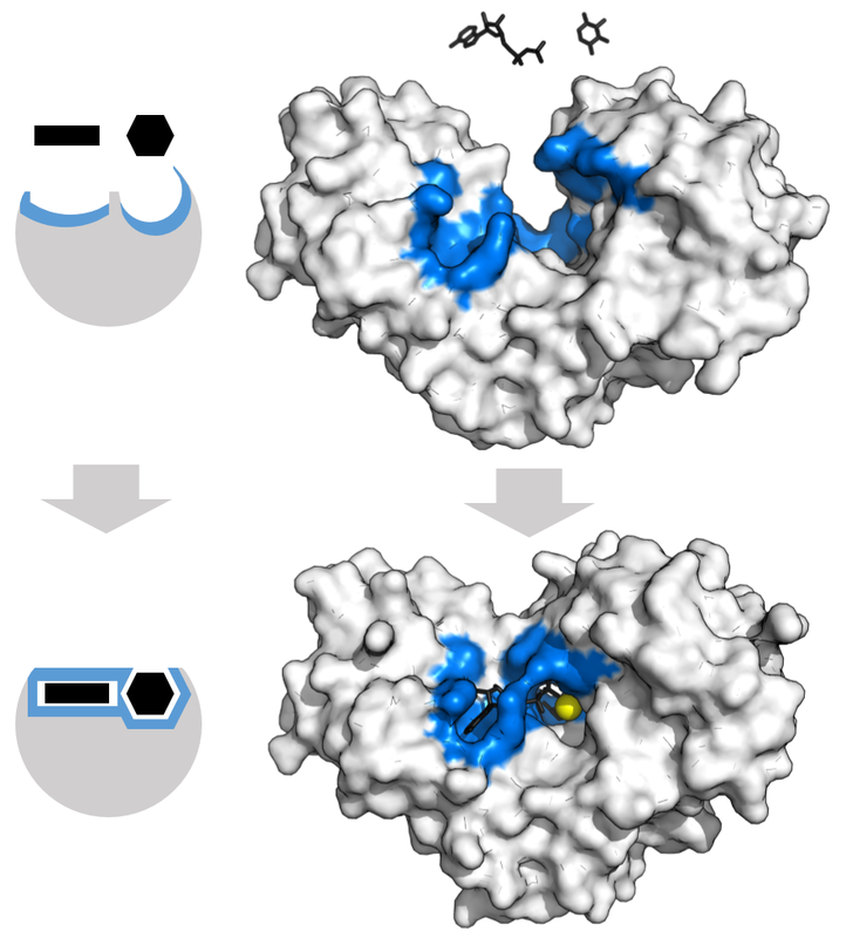
- Lock-and-key fit
- Highly regulated
- Initiate and/or speed up reactions (catalyze reactions and are referred to as biological catalysts)
- Require energy (endothermic)
- Release energy (exothermic)
- Metabolism
- May require an organic or inorganic cofactor, such as a metal ion, iron-sulfur, heme, or flavin
- May require a coenzyme, a small, organic molecule that may be bound either loosely or tightly to the enzyme, such as ATP, NADH, NADPH, a vitamin, or coenzyme A
- Results in a product or multiple products
- May be inhibited by a competitor, such as an antibiotic, poison, or other drug binding at an allosteric site and this may permanently alter or inactivate the enzyme
- Large, macromolecules with a 3D or 4D globular structure and shape
- Act on substrates and convert them to products
- Undergo a conformational change upon binding substrate
- Almost all metabolic activities in the body require enzymes to sustain life
- It's activity increases at ideal temperature and pH, and decreases at a less-than-ideal temperature and pH
- Used to break things down (catabolism) or build/synthesize new molecules (anabolism)
- Denature upon heating (unfold and lose their unique shape) or upon exposure to drugs or chemicals meant to disrupt their activity
- They have specific binding sites for substrates, with a catalytic site next to the binding site. Together these form the enzyme's active site.
- They are flexible and dynamic
- May be responsible for signal transduction
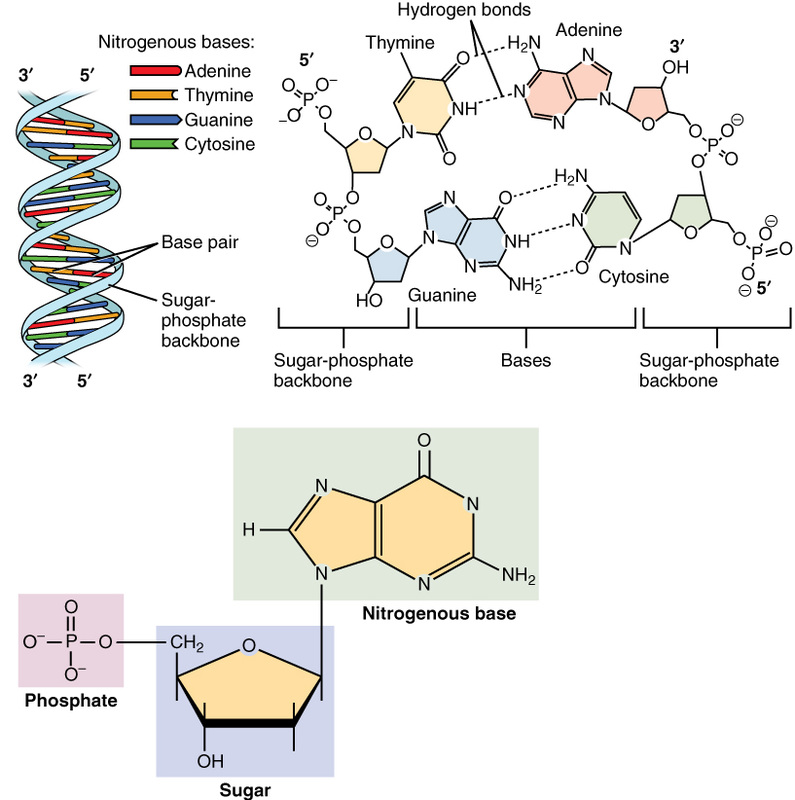
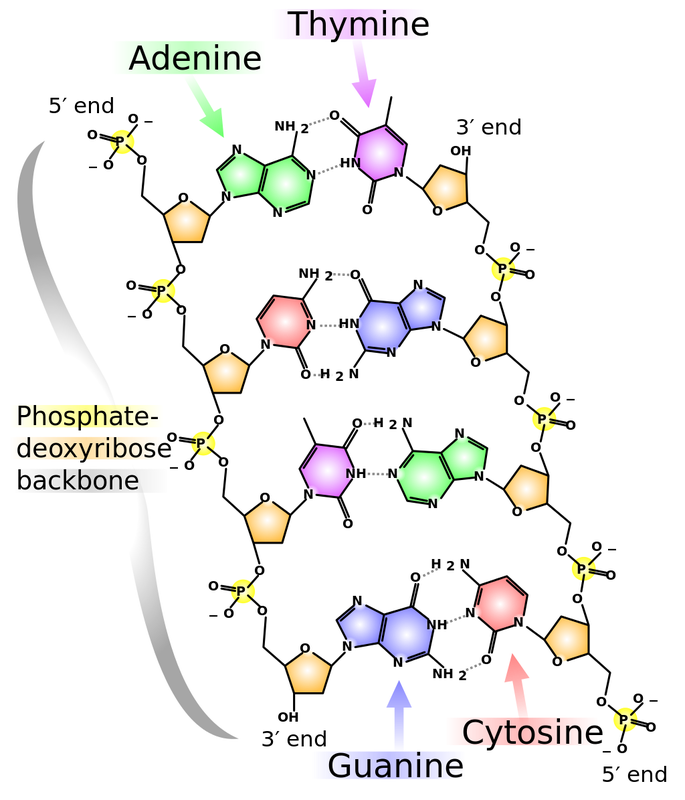
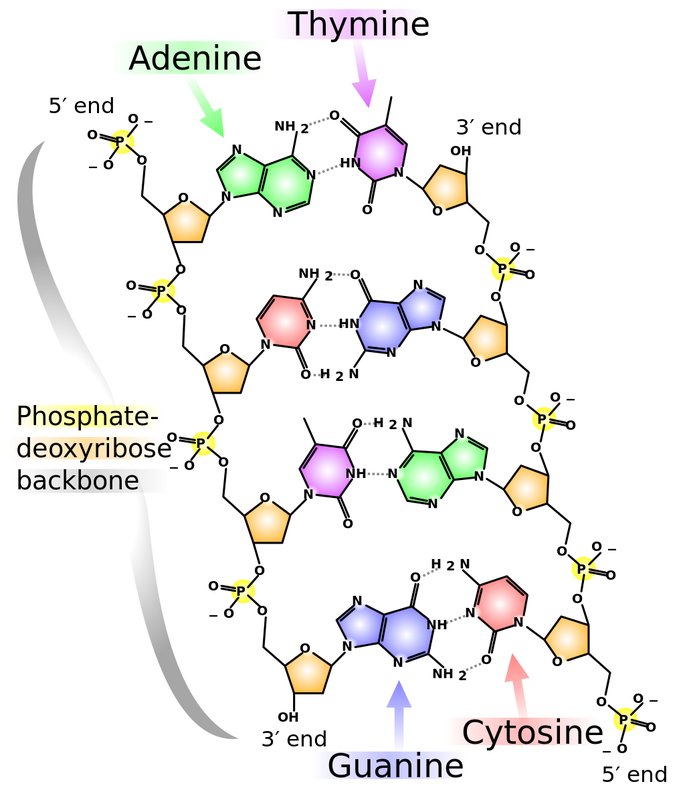
Deoxyribonucleic Acid: DNA
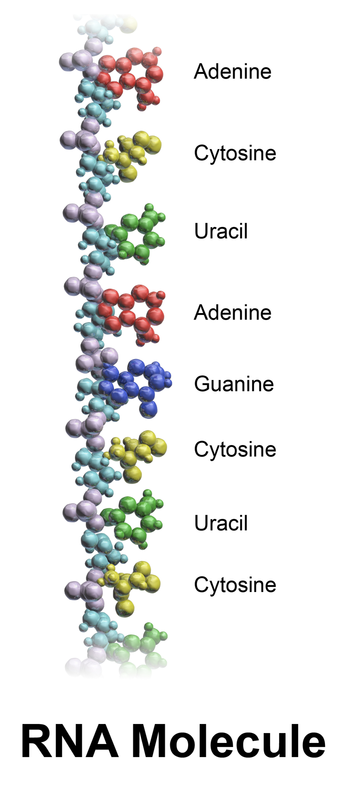
Ribonucleic Acid: RNA
Acids and Bases:
ACIDS:
- Below 7.0 on the pH scale
- Substances that increase the concentration of hydrogen (H+) ions when dissolved in water
- Substance which can act as a proton donor
- Substance which can accept a pair of electrons to form a covalent bond
- May be strong or weak
- Completely dissociate in water
- Hydrochloric acid (HCl) is a strong acid which is found in the gastric acid of the stomach and activates digestive enzymes
- Acetic acid is an example, found in vinegar
- Citric acid is an example, found in vitamin C and citrus fruits
- Sulfuric acid is an example, found in car batteries
- Tartaric acid, used in baking
- Derived from solids, liquids, or gases
- Boric acid is an example (borax detergent)
- Sour
- Turn litmus paper RED
- Able to react with bases
- Able to react with certain metals to form SALTS
- Include alcohols and amines, metal cations
- Strong acids are corrosive
- Some weak acids (concentrated) are corrosive
- Pickling
- Removing Rust
- Car Battery
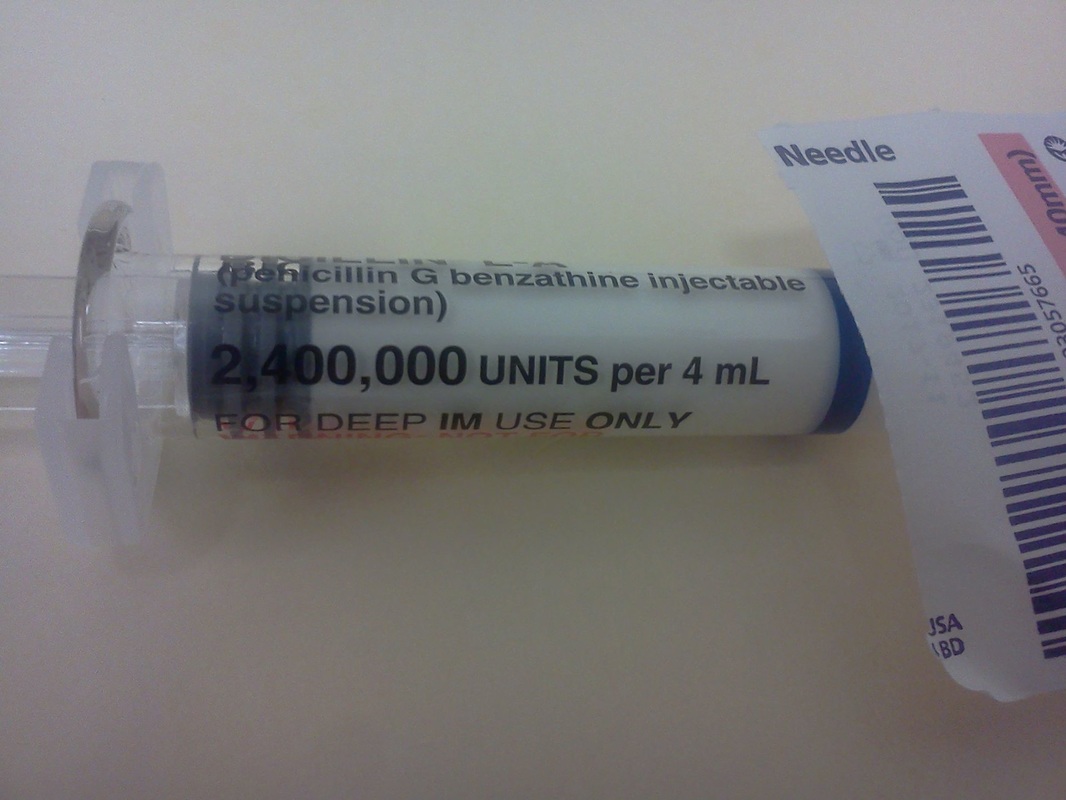
- Aspirin (acetylsalicylic acid) to treat fever/pain (weak acid)
- Ibuprofin (weak acid)
- Penicillin (weak acid; antibiotic)
- Cleaners
- Phosphate fertilizers
- Zinc oxide (diaper rash ointment, acne creams, skin healing)
- Additives to drinks and foods; Preservatives
- Oxalic acid (tomatoes, spinach, rhubarb) is an antioxidant and good for vision
- Ascorbic acid is found in vitamin C, and aids in immunity
- Amino acids (protein building blocks; growth and repair of tissue)
- Carbonic acid (pH homeostasis)
- Nucleic acids (building blocks of DNA and RNA)
- Antiseptics (mouth wash, bactericidals, topical medications)
- Insecticides
- Flame retardants
- Above 7.0 on the pH scale
- May be strong or weak
- Completely dissociate in water
- Sodium hydroxide is a strong base
- Alkaline
- Substances that, when in aqueous solution, are slippery, bitter
- Turn litmus paper BLUE
- React with acids to form SALTS
- Accept protons from a proton donor
- Produce hydroxide ions in aqueous solution
- It can accept hydrogen ions (H+), or protons
- It is an electron pair donor
- Strong or concentrated bases are caustic
- Strong bases react in a volatile manner with acids
- Molten bases dissociate in ions and conduct electricity
- Sodium hydroxide is a strong base
- Ammonia is a base
- Baking soda is a weak base
- Egg white is a weak base
- Potassium hydroxide is a base
- Sodium carbonate is a base
- Potassium fluoride is a base
- Carbon, nitrogen and oxygen can act as bases
- Other strong bases include: Lithium hydroxide, Sodium hydroxide, Rubidium hydroxide, Cesium hydroxide, Magnesium hydroxide, Strontium hydroxide, Barium hydroxide
- Serve as catalysts for chemical reactions
- Silica, Resins, Charcoal are bases
- Soaps
- Paper and pulps
- Rayon fabric
- Bleaching powder (hair dye, flour, paper, toilet paper, water treatment, tooth paste)-Calcium hypochlorite: Ca(ClO)2
- Antacids
- Water softeners
- Washing soda (laundry)
- Carpet cleaner
- Baking soda
- Baking powder
- Baby powder, talcum powders, facial powders
- Cooking
- Some types of fire extinguishers
- 7.0 on the pH scale
- Water is neutral
- These are buffers
Examples of Acids:
Examples of Bases (Alkaline):
The pH Scale:
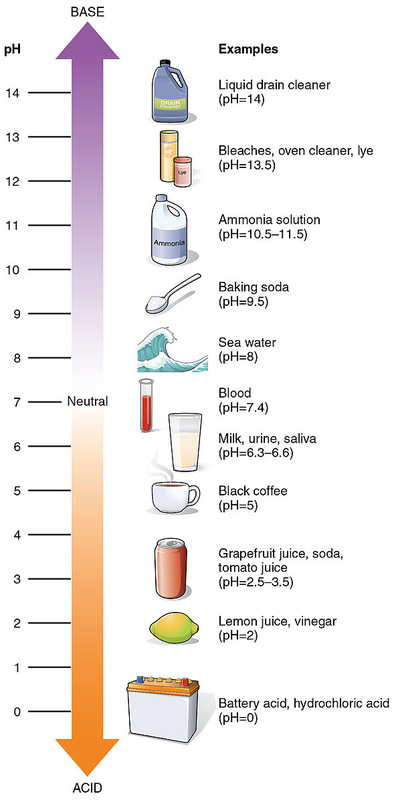
The pH scale is used in science and medicine as a numeric scale specifying the acidity or alkalinity of a solution and to measure the strngth of acids and bases (See image below). Indicators in the forms of strips or dyes are used to measure or detect pH.
On the pH scale, 7 is neutral, <7 is acidic, and >7 is alkaline or basic. The closer the pH is to 14, to stronger the base. The closer it is to just above 7, the weaker the base. The closer the pH is to 0, the stronger the acid. The closer it is to just under 7, the weaker the acid. Hydrochloric acid and battery acids are the strongest acids. Liquid drain cleaner, bleaches, oven cleaners and lye are the strongest bases.
The pH of blood is 7.35-7.45.
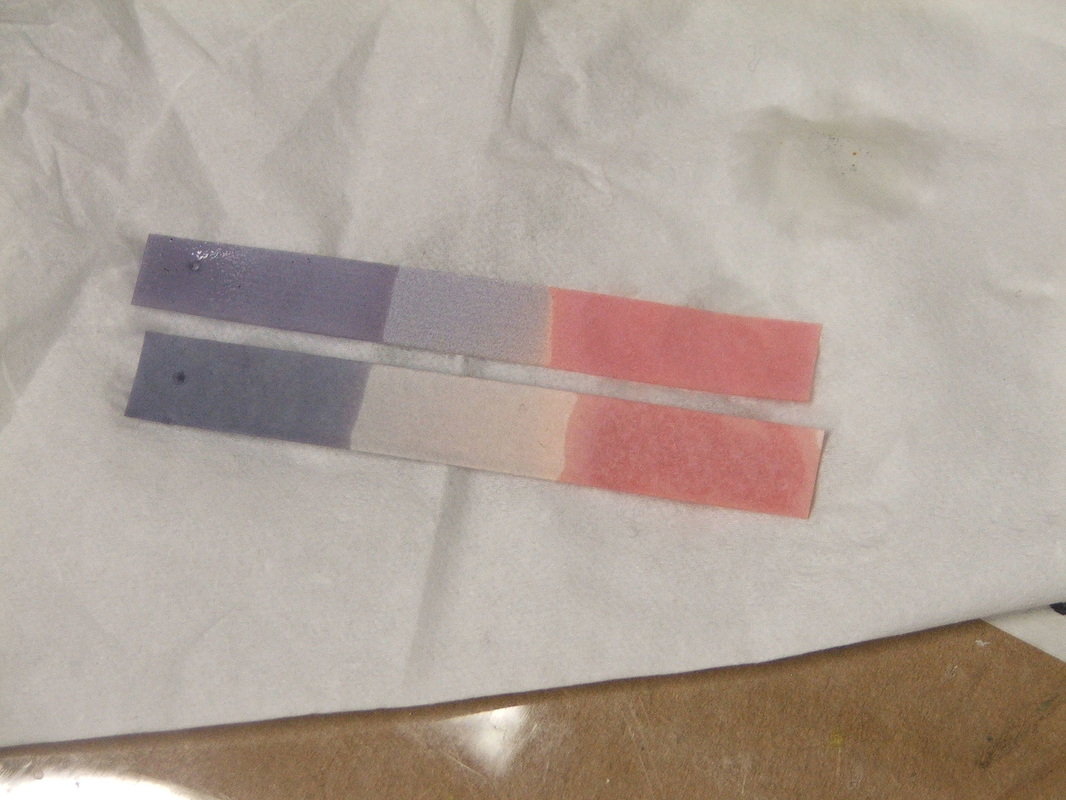
Whether or not a chemical or substance is acidic or basic/alkaline can be tested by a variety of methods. One includes litmus paper. Litmus paper is designed to test whether a substance is acidic or alkaline/basic, but it does not tell you exactly what number it is on the pH scale to be exact. It can only tell you if the substance is acidic or alkaline/basic, and this is associated with a pH color indicator on the paper. When litmus paper is dipped into an acidic substance, the red area stays red and the blue portion turns red. When litmus paper is dippped in a basic/alkaline substance, the blue portion stays blue, and the red portion turns blue (See image below).
On the pH scale, 7 is neutral, <7 is acidic, and >7 is alkaline or basic. The closer the pH is to 14, to stronger the base. The closer it is to just above 7, the weaker the base. The closer the pH is to 0, the stronger the acid. The closer it is to just under 7, the weaker the acid. Hydrochloric acid and battery acids are the strongest acids. Liquid drain cleaner, bleaches, oven cleaners and lye are the strongest bases.
The pH of blood is 7.35-7.45.
Whether or not a chemical or substance is acidic or basic/alkaline can be tested by a variety of methods. One includes litmus paper. Litmus paper is designed to test whether a substance is acidic or alkaline/basic, but it does not tell you exactly what number it is on the pH scale to be exact. It can only tell you if the substance is acidic or alkaline/basic, and this is associated with a pH color indicator on the paper. When litmus paper is dipped into an acidic substance, the red area stays red and the blue portion turns red. When litmus paper is dippped in a basic/alkaline substance, the blue portion stays blue, and the red portion turns blue (See image below).
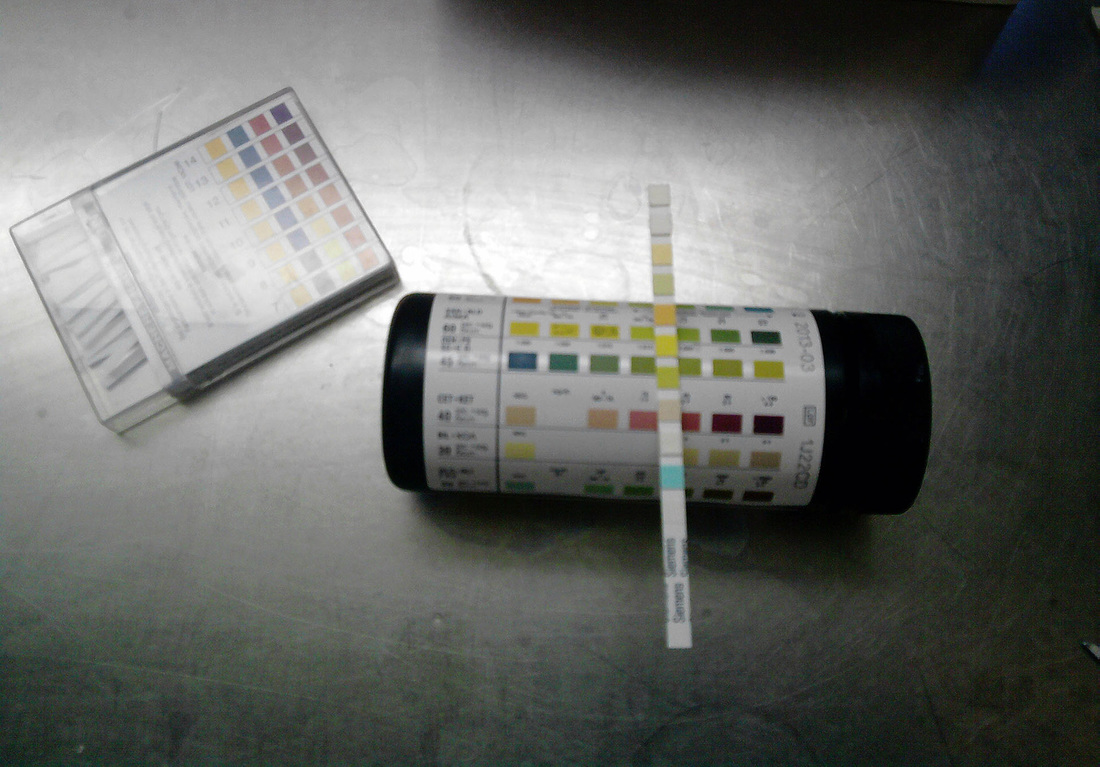
In laboratory medicine, understanding pH is important, because pH testing is used to test the pH levels in the stool, urine, and other body fluids to aid in the diagnosis of diseases, which may change or cause fluctuations in pH levels. Seen in the image above is an example of a urinalysis, where pH of urine is tested, in addition to other tests on the strip designed to detect the presence of red blood cells, white blood cells, sugars, or other substances in the urine that are not normal and aid in the diagnosis of urinary tract infections or other conditions in the body.
Buffers:
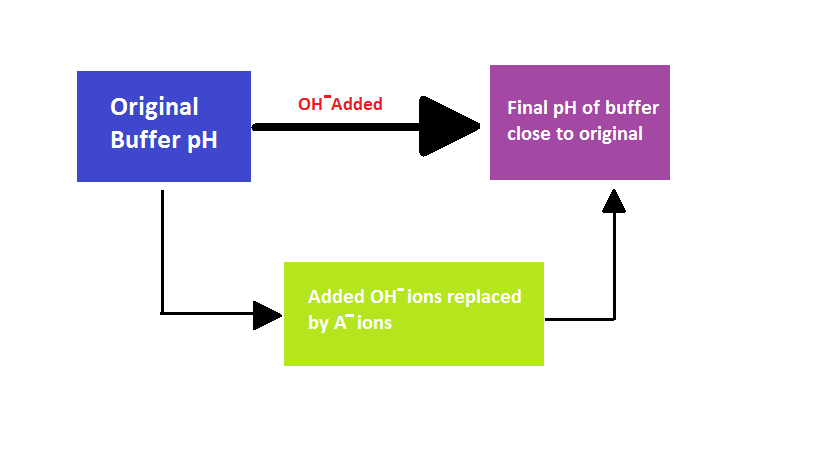
A buffer is basically either a base or weak acid, or a combination of both, used to maintain the pH of a solution and keep it in balance. In living organisms, buffering agents keep in the body in homeostasis or equilibrium. This is critical for life. An example of a buffer is bicarbonate, which maintains the pH of blood in the human body. Proteins inside cells, as well as phosphate, are also natural buffers.
Electrolytes:
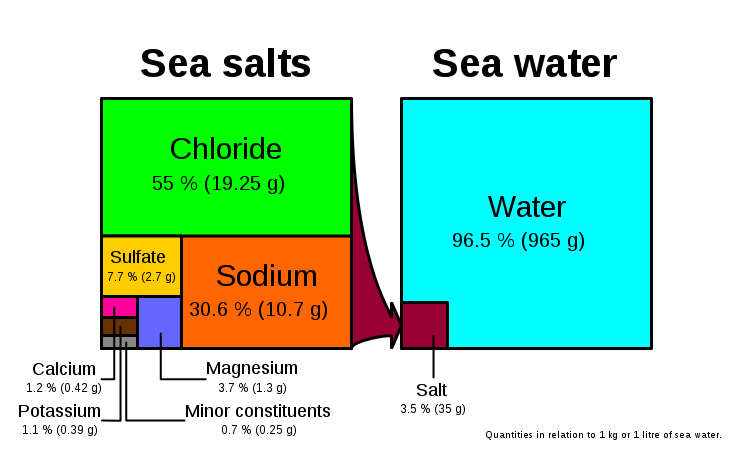
- These are ions that are able to conduct electricity in solution (acids, bases, salts)
- Sodium, magnesium, calcium, potassium, bicarbonate, chloride are all examples
- The body maintains the levels at appropriate ranges in order to maintain homeostasis and function properly
- Sports drinks and IV fluids are used to replace electrolytes lost during heavy exercise or vomiting/diarrhea
CASE STUDY: Written by Jeanette Reynolds, MS-Biology, M-ASCP, MLS-AMT, Certified in Emerging Diseases, Certified Instructor, Infection Preventionist
This is a practical, case study-style forum that isn't graded, but designed to help you interact with your peers while you discuss a couple of case studies, and use the terminology you learned last week to interpret the terminology and your healthcare provider role in the case.
Patient Record:
A patient arrived at the emergency room with complaints of RUQ pain, tenderness and swelling radiating to the epigastric region. A triage nurse called the patient back to be assessed and initial vital signs were taken (temperature and blood pressure, pulse, pulse oxygen, and respiratory rates) and signs and symptoms were documented. The nurse noted a buildup of ascites fluid in the abdominal cavity. The temperature revealed a low-grade fever. The blood pressure and pulse were slightly elevated, but all other values were normal.
Another individual came in and obtained her insurance information and patient history and documented it. A phlebotomist came in a drew blood for lab work, which was quickly sent to the lab, where tests were performed by the MLT's and MLS's. Initial labs indicated an increase in white blood cells (leukocytosis), indicating potential inflammation and/or infection. Other lab results remained pending. A P.A. in the ER assessed the patient, palpated the RUQ, which triggered pain and tenderness, and they suspected swelling in this area, so they ordered an X-ray first, both in standard anatomical position and in supine position, which did not reveal any abnormalities and appeared unremarkable. A CT-scan with and without contrast (dye made of radioactive isotopes) was ordered, both in supine position, which may reveal more detail. The CT-scan with contrast results came back indicating a suspicious lesion with dysplasia.
A surgical biopsy was scheduled for the following day. The patient was prepared in supine position and a nurse anesthesiologist administered general anesthesia. The patient's skin over the RUQ was prepared and cleansed with antiseptic iodine (surgical scrub). A surgical cut was made in the RUQ in order to obtain a liver biopsy. The biopsy results were ordered STAT, and the results revealed hepatomegaly and carcinoma secondary to cirrhosis. The pending lab results appeared the next day, which were positive for Hepatitis C, a virus linked to liver cirrhosis and cancer.
Questions:
- What is the name of the organ(s) potentially involved in this scenario?
- What does "RUQ" stand for? What organs might be located in this area?
- Which abdominal regions are affected?
- Which body planes could you view this scenario in?
- What body cavity or cavities would this organ lie in?
- Which abdominal regions does it lie in?
- What could you see your role in this particular scenario? What parts would you play?
- How would describe the "supine" position?
- How would you describe the standard anatomical position used for the X-ray in the scenario?
- Which region and quadrant is the surgical cut being made in?
- If they decide to do further testing on the ascites fluid buildup or remove it (called an abdominocentesis), which cavities would they be pulling the fluid out of?
- What is an organ that the liver is superior to? What is an organ that it is inferior to? Is it bilateral?