Histology: The Study of Tissues
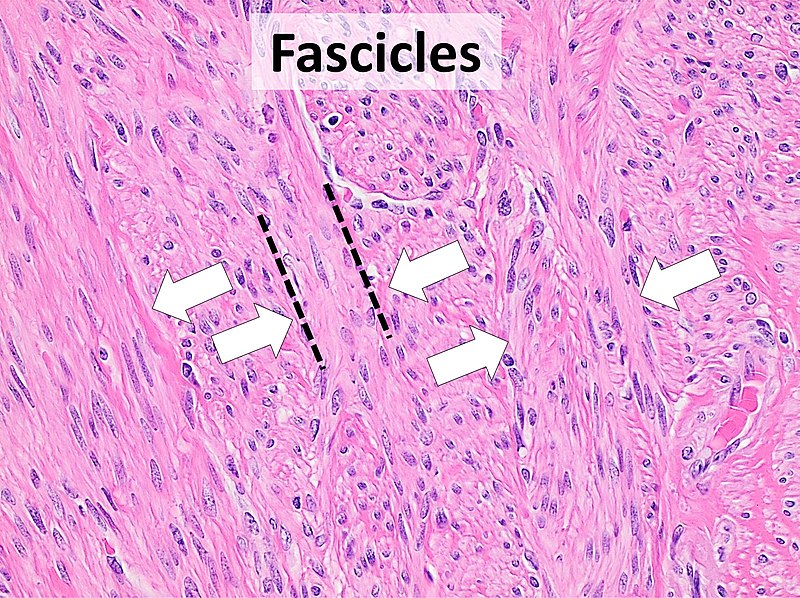
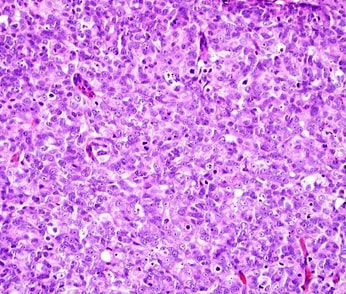
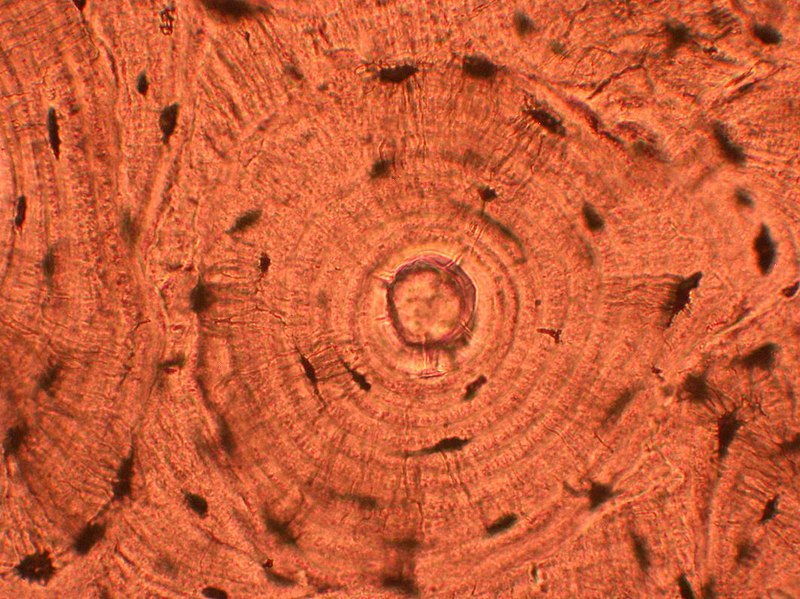
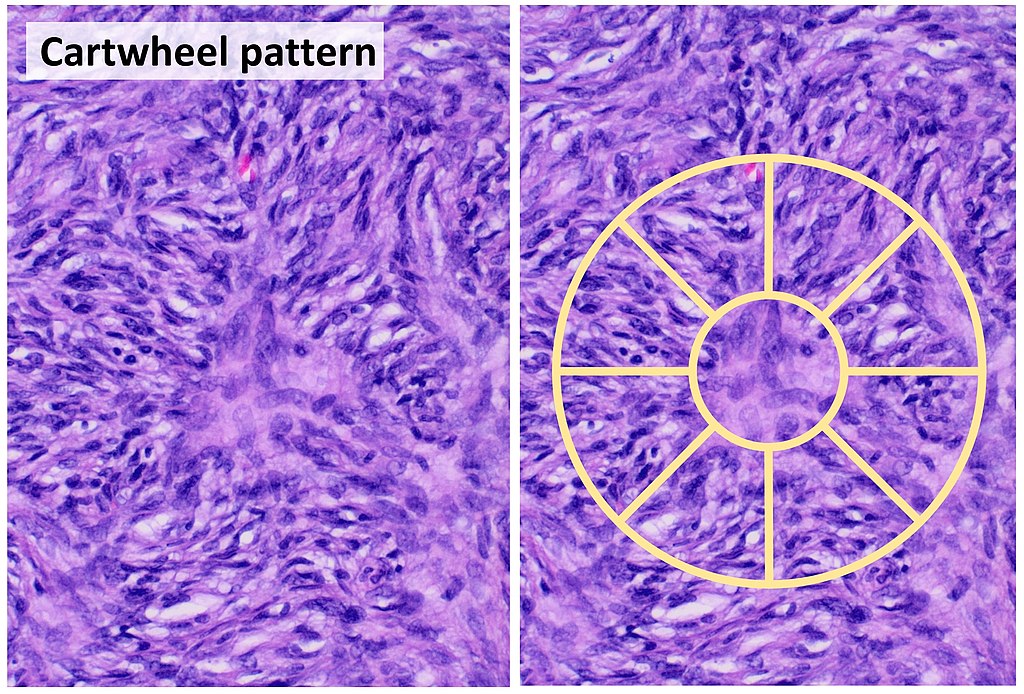
Also known as "microscopic anatomy" or "microanatomy", histology is the study of the structure of the tissues of the human body. "Gross anatomy", or "macroscopic anatomy", is the opposite, being the study of the larger tissues seen with the naked eye. Histopathology is included in this branch of study, including the observation of diseased tissues, not just healthy ones. This aids pathologists in the identification of diseases.
There are four major types of tissues, as you've learned and can read about in the tissues tab. They include:
Medical histologists are Medical Lab Scientists that work in the Histology section of the lab under the supervision of a licensed Pathologist (MD or MD/PhD), prepare the samples to fix and stain them to make permanent slides, and the Pathologist reviews them and looks at them to differentiate them and identify diseases. There are two branches: anatomical pathology and surgical pathology. They identify normal tissue, diseased tissue, lesions, tumors (benign or malignant), including cancers.
Fixatives are special chemicals used to preserve specimens and to keep the cells and tissues intact so they can be examined and made into slides. Fixation also makes the tissues hard enough that they can be cut into super thin microscopic sections to be molded onto a slide and stained for microscopic viewing. The most common fixative used is 10% formalin, or 4% formaldehyde in phosphate buffered saline.
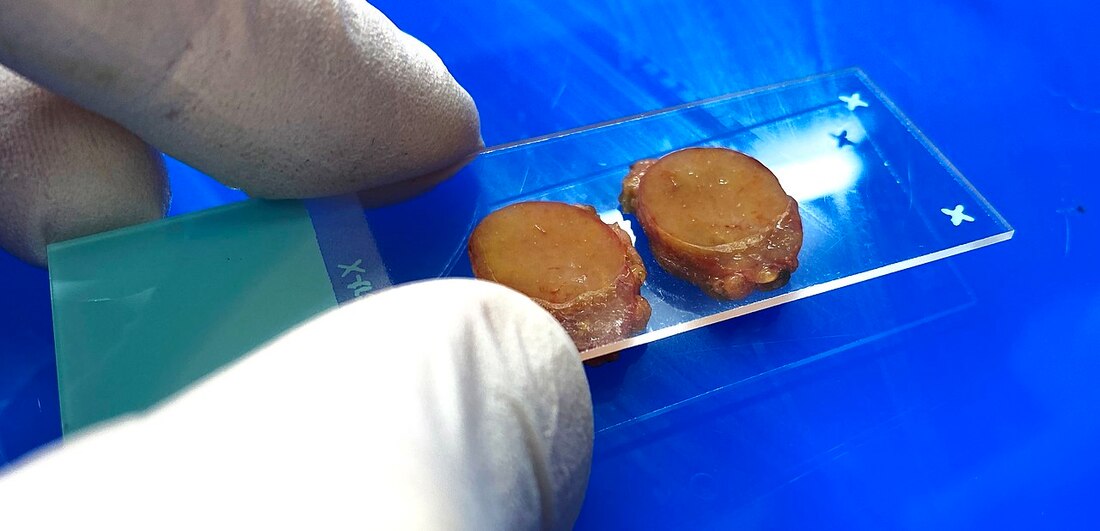
Tissues are observed and analyzed and only areas of suspicious or inflamed tissue are needed, and the rest is removed and cut off. This step is known as "selection" and "trimming". In some cases, the entire tissue is needed, as in the case of certain glands, lymph nodes, or moles, so "selection" is the only step needed first. The second step is known as "dehydration" and "embedding". The dehydration step involves allowing the tissues to dry out a little bit through dipping them in a series of progressively concentrated ethanol solutions, which removes excess water, then using a clearing fluid, often xylene, to get rid to remove excess debris and act as a surfactant to bind the tissue with the embedding medium in the next step. The embedding step typically uses melted paraffin wax, which stiffens up the tissues enough to be cut into very thin slices by a special machine called a microtome. Some laboratories have automated tissue processing machines that complete these processes rapidly. Tissues are thin dipped in paraffin wax, then placed into wax-filled molds, cooled, and solidified into blocks. Finally, tissues are cut in a "frozen" state (from the blocks) by being dunked into a water-based embedding medium. Pre-frozen tissues are embedded in molds with liquid embedding material and then frozen.
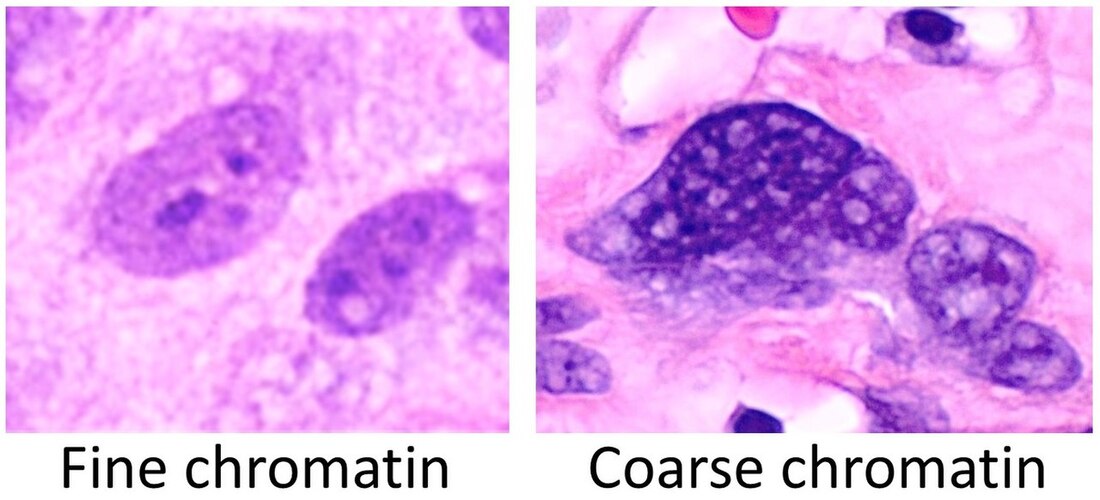
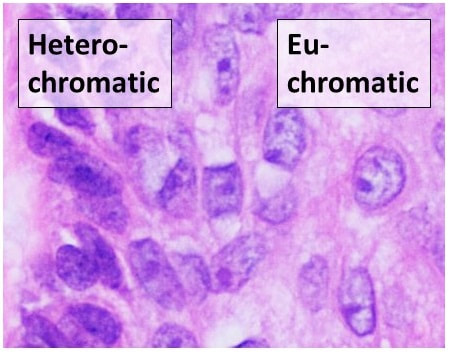
The next process is known as "sectioning". A microtome is a special type of machine that contains a very sharp blade that cuts the blocks into very thin slices just 5-15 micrometers thick, which are mounted onto clean, glass microscopy slides. Cryosectioning is also a special type of procedure that can be performed in order to quickly freeze, cut and mount sections of tissue, and it still uses a microtome, but it is a freezing microtome, or a cryostat. The frozen sections are mounted on the slides and can also be stained. This is helpful for certain surgeries, such as Mohs surgery, in which a suspicious tumor is removed, along with a margin of healthy tissue surrounding it, to determine whether or not it is malignant. Once this step has been completed, the slides are ready to be stained, and there are a variety of stains that can be used. This will provide contrast to the tissue, and cause internal features to stand out. Some of the stains that are used are as follows:
Pathologists analyze the slides, and then generate a pathology report, which is sent to the patient's physician, who will then contact them with the results.
There are four major types of tissues, as you've learned and can read about in the tissues tab. They include:
- Epithelial tissue
- Muscle tissue
- Nervous tissue
- Connective tissue
Medical histologists are Medical Lab Scientists that work in the Histology section of the lab under the supervision of a licensed Pathologist (MD or MD/PhD), prepare the samples to fix and stain them to make permanent slides, and the Pathologist reviews them and looks at them to differentiate them and identify diseases. There are two branches: anatomical pathology and surgical pathology. They identify normal tissue, diseased tissue, lesions, tumors (benign or malignant), including cancers.
Fixatives are special chemicals used to preserve specimens and to keep the cells and tissues intact so they can be examined and made into slides. Fixation also makes the tissues hard enough that they can be cut into super thin microscopic sections to be molded onto a slide and stained for microscopic viewing. The most common fixative used is 10% formalin, or 4% formaldehyde in phosphate buffered saline.
Tissues are observed and analyzed and only areas of suspicious or inflamed tissue are needed, and the rest is removed and cut off. This step is known as "selection" and "trimming". In some cases, the entire tissue is needed, as in the case of certain glands, lymph nodes, or moles, so "selection" is the only step needed first. The second step is known as "dehydration" and "embedding". The dehydration step involves allowing the tissues to dry out a little bit through dipping them in a series of progressively concentrated ethanol solutions, which removes excess water, then using a clearing fluid, often xylene, to get rid to remove excess debris and act as a surfactant to bind the tissue with the embedding medium in the next step. The embedding step typically uses melted paraffin wax, which stiffens up the tissues enough to be cut into very thin slices by a special machine called a microtome. Some laboratories have automated tissue processing machines that complete these processes rapidly. Tissues are thin dipped in paraffin wax, then placed into wax-filled molds, cooled, and solidified into blocks. Finally, tissues are cut in a "frozen" state (from the blocks) by being dunked into a water-based embedding medium. Pre-frozen tissues are embedded in molds with liquid embedding material and then frozen.
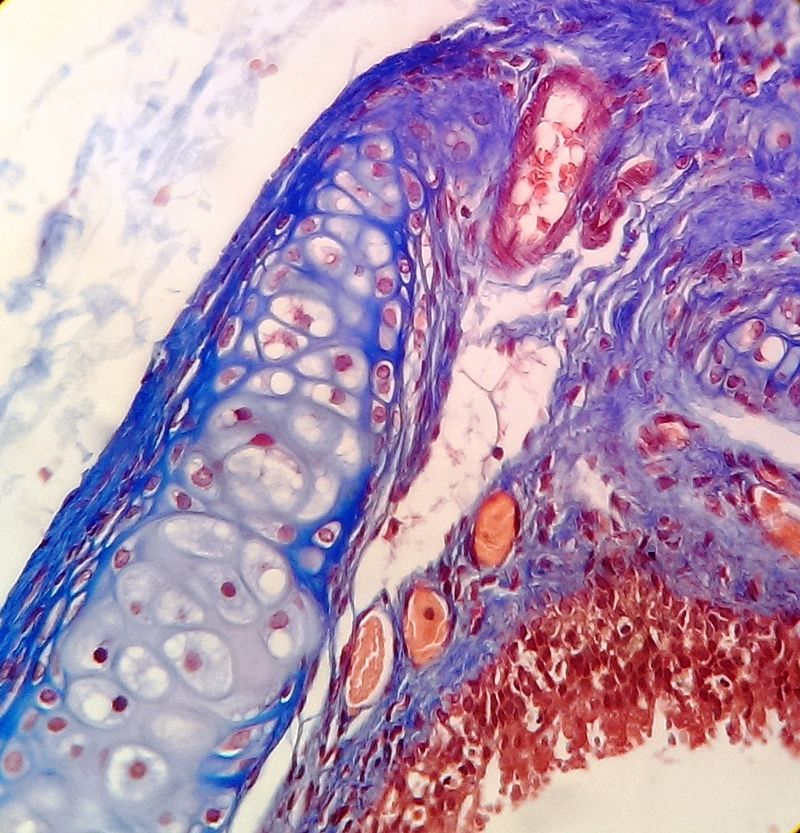
The next process is known as "sectioning". A microtome is a special type of machine that contains a very sharp blade that cuts the blocks into very thin slices just 5-15 micrometers thick, which are mounted onto clean, glass microscopy slides. Cryosectioning is also a special type of procedure that can be performed in order to quickly freeze, cut and mount sections of tissue, and it still uses a microtome, but it is a freezing microtome, or a cryostat. The frozen sections are mounted on the slides and can also be stained. This is helpful for certain surgeries, such as Mohs surgery, in which a suspicious tumor is removed, along with a margin of healthy tissue surrounding it, to determine whether or not it is malignant. Once this step has been completed, the slides are ready to be stained, and there are a variety of stains that can be used. This will provide contrast to the tissue, and cause internal features to stand out. Some of the stains that are used are as follows:
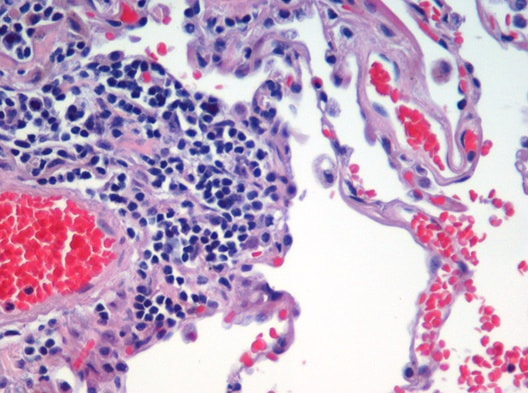
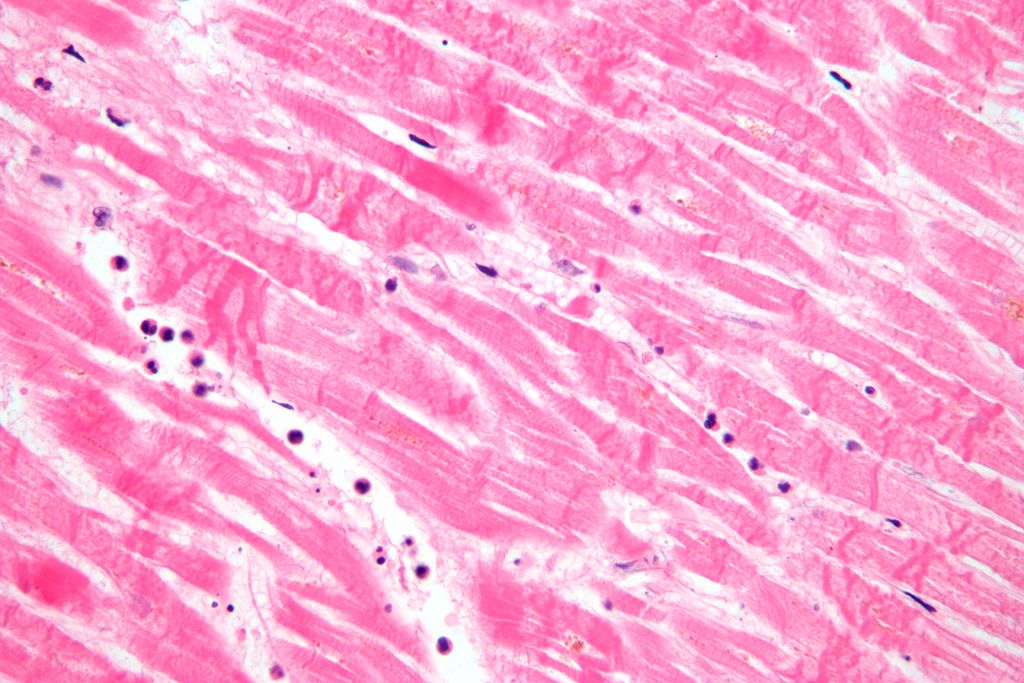
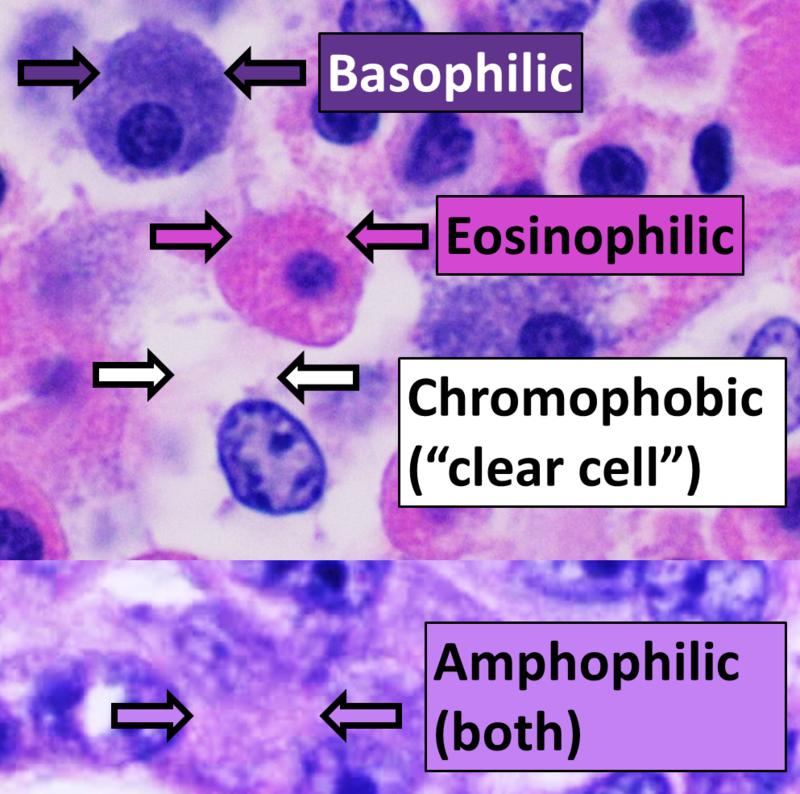
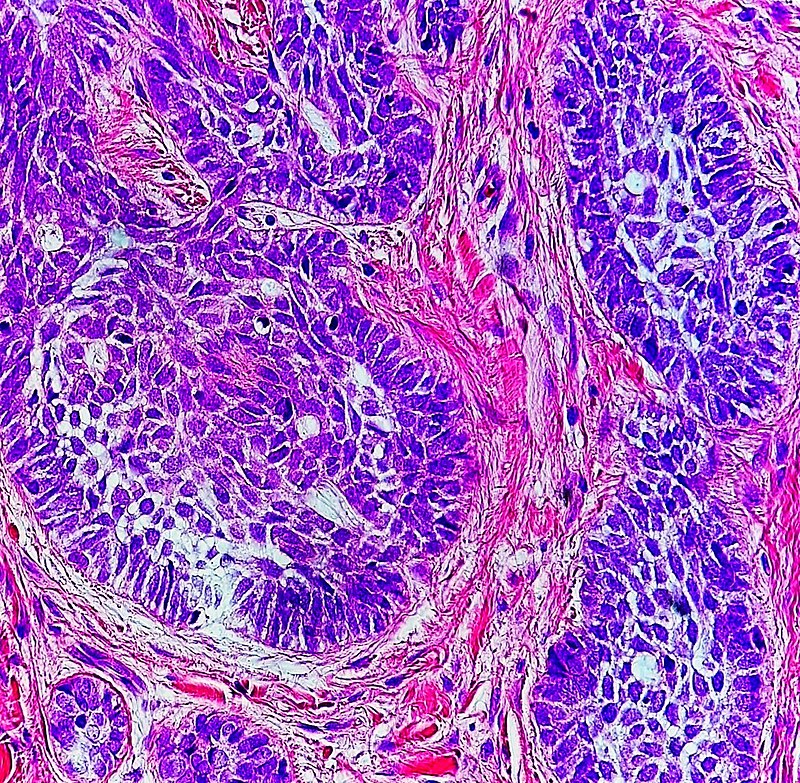
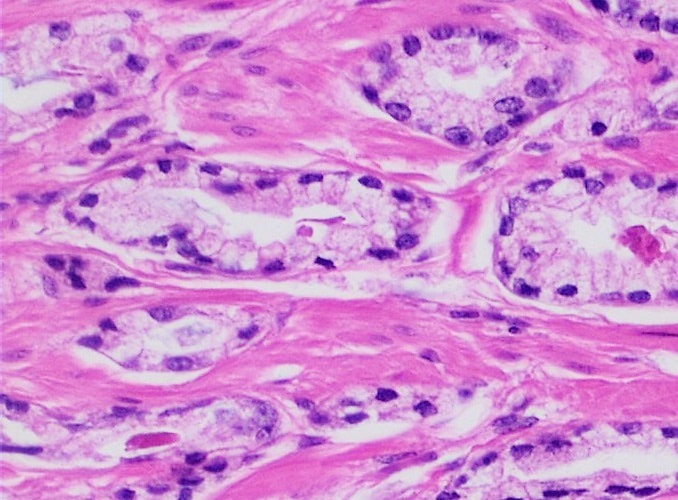
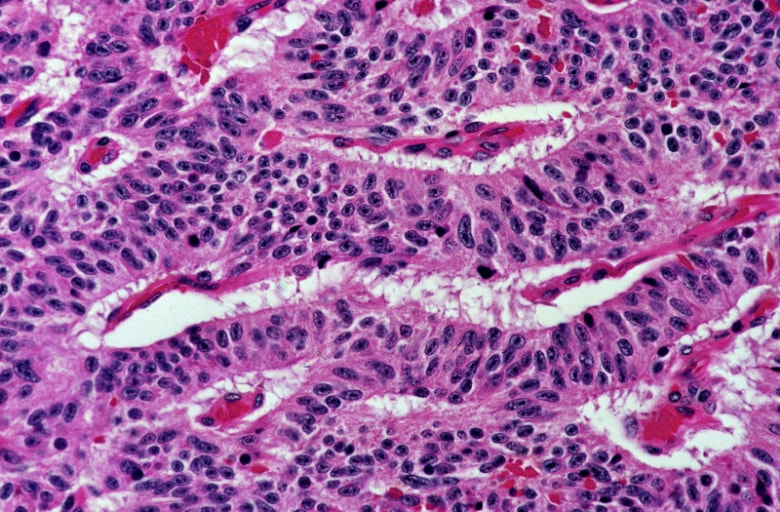
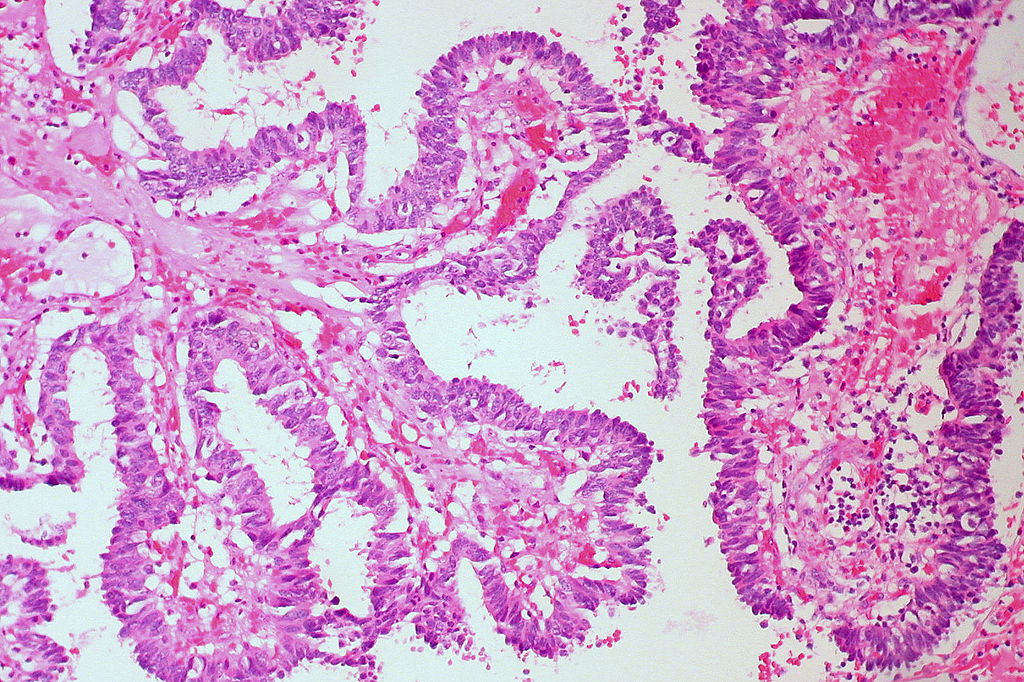
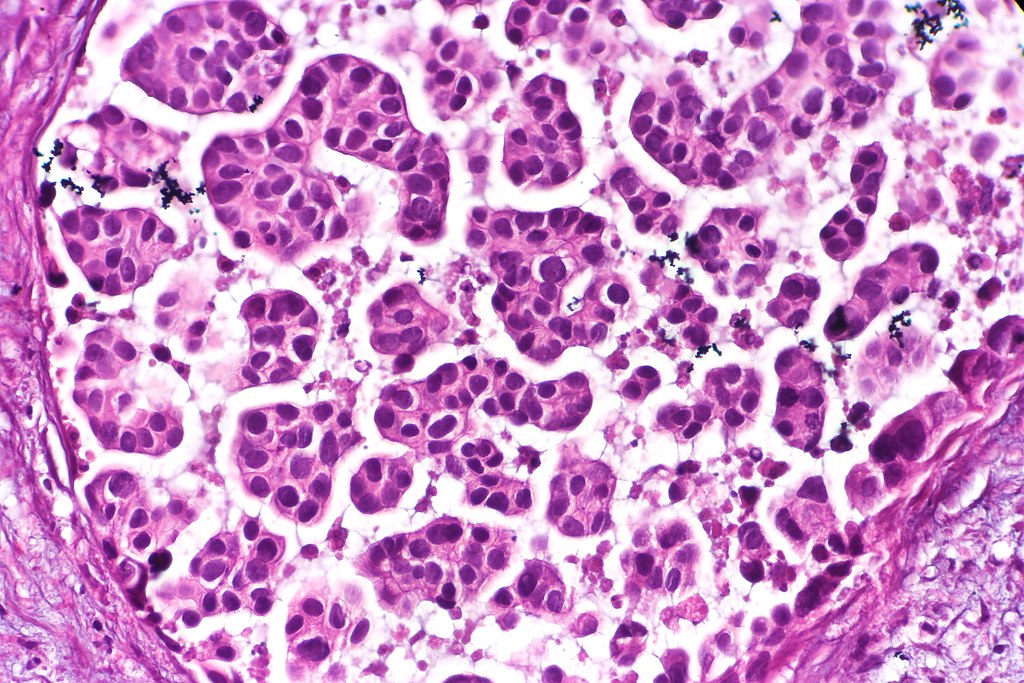
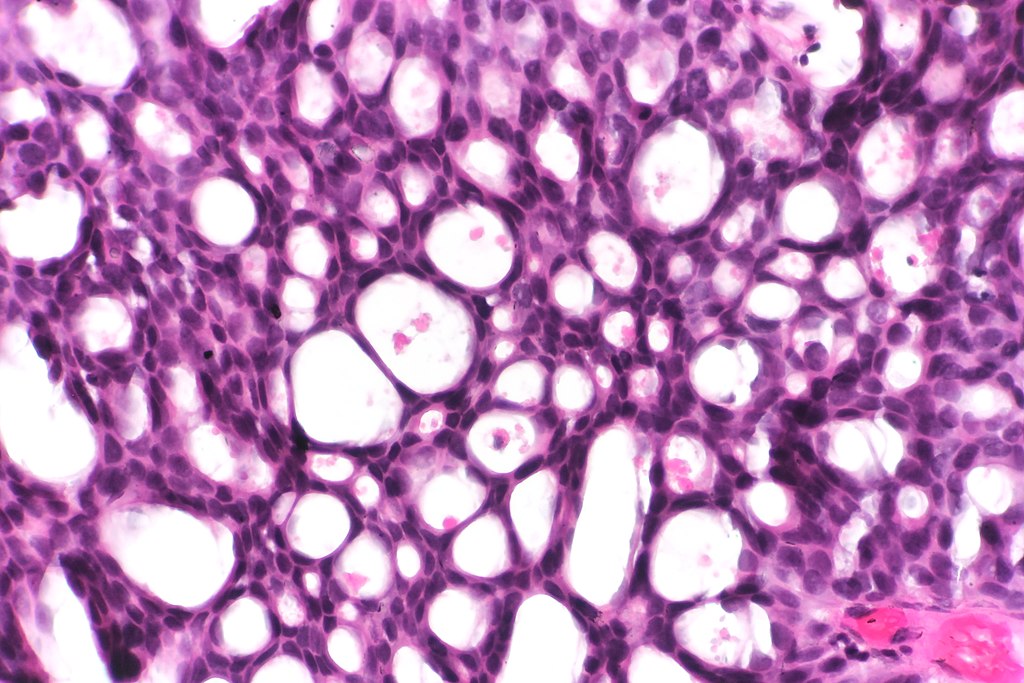
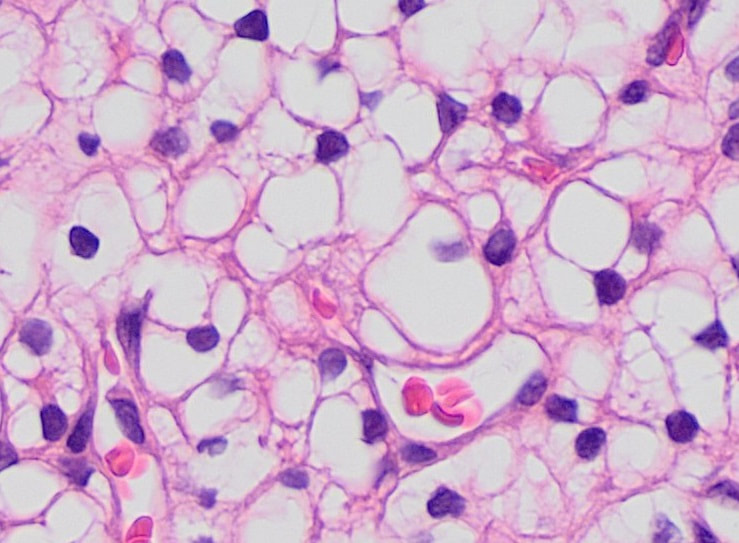
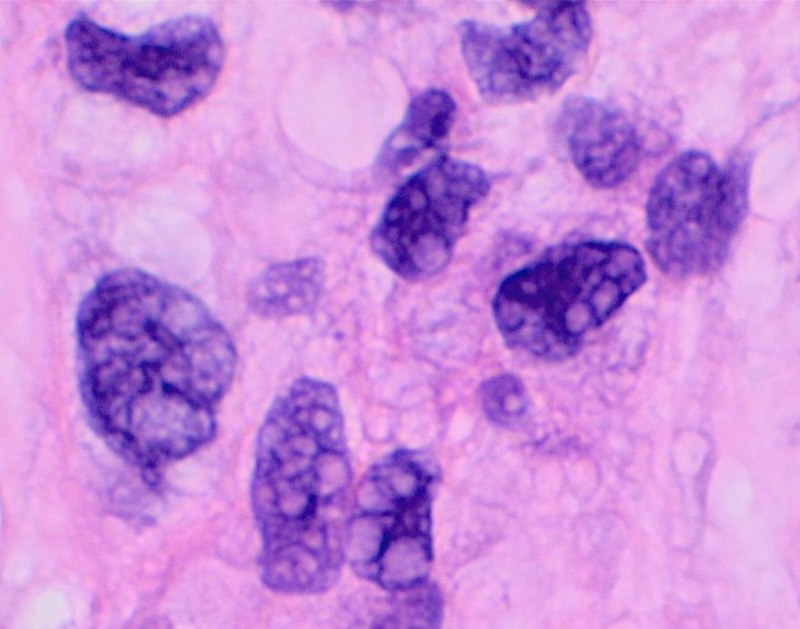
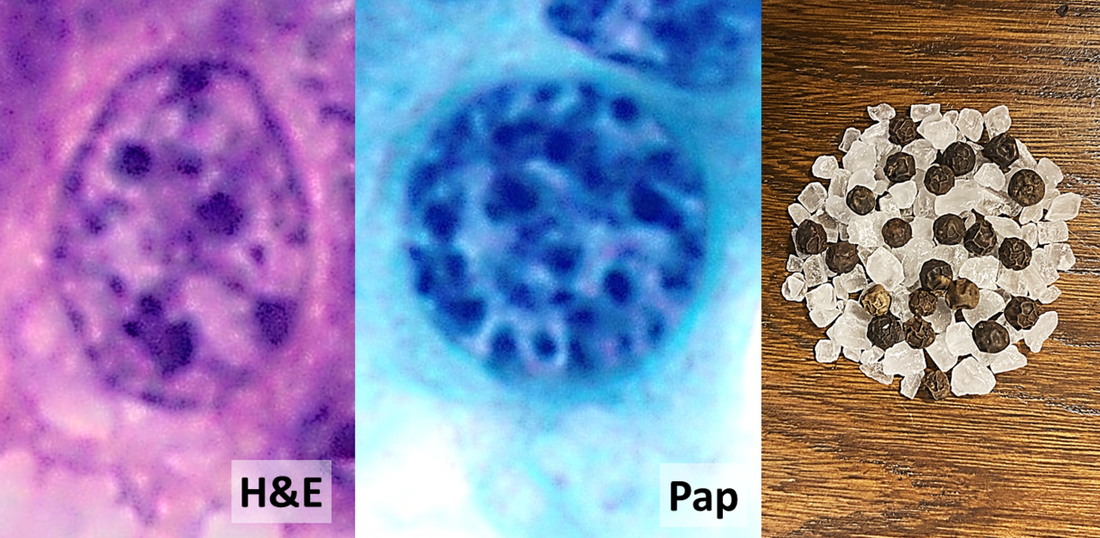
- Hematoxylin and Eosin (H&E) Stain: This is one of the most common stains. Hematoxylin enables the nucleus to stain blue, whereas Eosin enables the cytoplasm and other structures to stain pink. I personally think this is one of the prettiest stains in histology. Eosin is an acidic dye.
- Prussian Blue: This stain is used to stain iron deposits that might be present in cells. This helps determine diseases like hereditary hemochromatosis.
- Nissl Method: This stain is used to stain neurons, neuroglia, and sections of the brain and spinal cord to look at nerves. Golgi's method and silver stains are also beneficial for staining of these structures.
Pathologists analyze the slides, and then generate a pathology report, which is sent to the patient's physician, who will then contact them with the results.